Complete genetic and epigenetic architecture of D4Z4 macrosatellites in FSHD, BAMS, and reference cohorts with D4Z4End2End
- Lucinda C. Xiao1,
- Ayush Semwal1,
- Brianna St John1,
- Kathleen Zeglinski1,
- Shian Su1,2,
- James Lancaster1,
- Shifeng Xue3,
- Bruno Reversade4,
- Matthew E. Ritchie1,2,
- Frédérique Magdinier5,
- Marnie E. Blewitt1,2 and
- Quentin Gouil1,2,6,7
- 1The Walter and Eliza Hall Institute of Medical Research, Parkville, VIC 3052, Australia;
- 2Department of Medical Biology, The University of Melbourne, Parkville, VIC 3052, Australia;
- 3Department of Biological Sciences, National University of Singapore, 117558, Singapore;
- 4Laboratory of Human Genetics and Therapeutics, King Abdullah University of Science and Technology, Thuwal 23955, Kingdom of Saudi Arabia;
- 5Marseille Medical Genetics, Aix-Marseille University, INSERM, Marseille 13005, France;
- 6Olivia Newton-John Cancer Research Institute, Heidelberg, VIC 3084, Australia;
- 7School of Cancer Medicine, La Trobe University, Bundoora, VIC 3086, Australia
Abstract
The D4Z4 locus is a macrosatellite array on Chromosome 4q normally comprising 8 to >100 3.3-kb repeat units. Its size and repetitiveness render it refractory to most sequencing technologies; consequently, its genetic and epigenetic architectures remain incompletely understood despite their relevance to facioscapulohumeral muscular dystrophy (FSHD). Current FSHD molecular testing relies on complex, multistep and low-resolution assays, which aim to identify contractions on permissive haplotypes (FSHD type 1) or epigenetic reactivation due to pathogenic variants in the epigenetic machinery, most often in SMCHD1 (FSHD type 2). Recent guideline updates highlight the need for more accurate and comprehensive diagnostic approaches. Here, we leverage ultra-long whole-genome and Cas9-targeted sequencing to develop a fast and accurate workflow, D4Z4End2End, for comprehensive genetic and methylation analysis of D4Z4 alleles. We apply it to samples from two controls, four FSHD1 patients, four FSHD2 patients, and two patients with Bosma arhinia microphthalmia syndrome (BAMS) caused by SMCHD1 variants, as well as publicly available data from 30 B-lymphoblastoid cell lines from the 1000 Genomes Project and Human Pangenome Reference Consortium. We attain high-depth sequencing of full-length D4Z4 arrays of up to 40 repeat units (∼132 kb), accurately capture contracted arrays, genetic mosaicism, and pathogenic SMCHD1 variants, and generate consensus sequences of all D4Z4 alleles. We identify new allelic variants, analyze complex D4Z4 rearrangements including in-cis duplications, and reveal length- and SMCHD1-dependent methylation patterns across the D4Z4 array. Our findings offer insights into D4Z4 genetics and epigenetics, and demonstrate the potential of long-read nanopore sequencing to accelerate FSHD research and diagnostics.
Facioscapulohumeral muscular dystrophy (FSHD) is a genetic disorder that causes progressive weakness of the muscles of the face, scapula and upper arms, as well as the lower legs, hip girdle, and abdomen (Preston and Wang 2025). It is the third most common muscular dystrophy worldwide, with an estimated prevalence of between 1/8000 and 20,000 (Mostacciuolo et al. 2009; Deenen et al. 2014; Wang et al. 2022).
FSHD has a complex etiology involving genetic and epigenetic dysregulation of the D4Z4 macrosatellite array in the subtelomere of Chromosome 4 (4q35). The D4Z4 array comprises 1 to >100 3.3-kb tandem repeats, each of which contains a partial copy of the DUX4 gene, with a full-length DUX4 gene being present at the distal end of the array (Fig. 1A). DUX4 encodes a transcription factor that is normally expressed in a small window of embryonic development, where it plays a key role in zygotic genome activation (De Iaco et al. 2017), but is thereafter silenced in most somatic tissues (Mocciaro et al. 2021). FSHD arises when loss of silencing at D4Z4 leads to aberrant expression of DUX4 in skeletal muscle, and subsequent activation of myotoxic signaling pathways (Campbell et al. 2018; Duranti and Villa 2023).
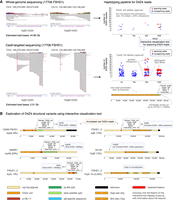
D4Z4End2End: an all-in-one, long-read-based workflow for the genetic and epigenetic analysis of FSHD. (A) Schematic of the pathogenesis of FSHD. The causative locus of FSHD is the D4Z4 macrosatellite array at the Chromosome 4q subtelomere, which comprises 1 to >100 ∼3.3 kb tandem repeat units. FSHD occurs in the presence of (i) a PAS-containing D4Z4 haplotype (4qA), which enables stable DUX4 expression and (ii) epigenetic dysregulation of the array, which can occur via array contraction (FSHD1) or pathogenic variants in chromatin modifiers (FSHD2). Both FSHD1 and FSHD2 are associated with D4Z4 hypomethylation and ectopic DUX4 expression in skeletal muscle. A nonpathogenic, homologous D4Z4 array is located at the Chromosome 10q subtelomere. A 4q-specific XapI site and 10q-specific BlnI site is used to distinguish 4q and 10q alleles in Southern blot assays. (B) Overview of the all-in-one, long-read-based workflow for FSHD. Ultra-high-molecular-weight (UHMW) DNA is used to perform Oxford Nanopore Technologies (ONT) Cas9-targeted or whole-genome sequencing (WGS). Canonical and 5mCG basecalling is performed using Dorado. Variants in reads overlapping FSHD2-associated genes are called using DeepVariant (Poplin et al. 2018), and the potential pathogenicity of these variants is assessed using Variant Effect Predictor (VEP) (McLaren et al. 2016). For assessment of the D4Z4 array, raw reads are input into a custom script which annotates and haplotypes D4Z4 reads (Supplemental Code). The output of the haplotyping pipeline can be explored using an interactive visualization tool, which displays raw reads as individual dots grouped into their haplotypes and plotted based on their number of D4Z4 repeat units (y-axis), and highlights 4q and 10q spanning reads. Hovering over individual dots displays the annotated raw read. Accurate consensus sequences for 4q and 10q alleles can be generated from spanning reads using Racon (Vaser et al. 2017), enabling analysis of allele-specific D4Z4 genetic variants. Allele-specific, array-wide methylation analysis is performed using NanoMethViz (Su et al. 2021, 2025), allowing detection and visualization of FSHD1- and FSHD2-specific methylation patterns.
Epigenetic derepression of D4Z4 can occur through two different mechanisms, which define the two main subtypes of FSHD (FSHD1 and FSHD2) (Fig. 1A) (Himeda and Jones 2019; Schätzl et al. 2021; Caputo et al. 2022b). FSHD1 accounts for the majority of cases (∼95%), and is caused by contraction of the D4Z4 array to 1–10 repeat units, which can either be inherited in an autosomal dominant manner or arise de novo (10%–30% of cases) (van der Maarel et al. 2000; Lemmers et al. 2004). FSHD2 (∼5% of cases) is caused by pathogenic variants in chromatin modifiers including SMCHD1 (Lemmers et al. 2012), DNMT3B (van den Boogaard et al. 2016), and LRIF1 (Hamanaka et al. 2020) in the presence of an intermediate-length array of commonly 8–20 repeat units (Giardina et al. 2024) or cis-duplicated arrays with short distal arrays (Lemmers et al. 2018a). Noncontracted D4Z4 arrays generally have high levels of DNA methylation, whereas both FSHD1 and FSHD2 are associated with hypomethylation of either the contracted array (FSHD1) or all D4Z4 alleles (FSHD2) (de Greef et al. 2009; De Greef et al. 2010; Gould et al. 2021; Erdmann et al. 2023).
Further factors complicate the genetics of FSHD (Fig. 1A). Two main haplotypes of the 4q D4Z4 allele have been identified, designated 4qA and 4qB, based on differences in the sequence immediately distal to the D4Z4 array (Lemmers et al. 2002). Only the 4qA haplotype contains a polyadenylation signal (PAS) required for stabilization of the DUX4 mRNA, and thus only 4qA alleles are pathogenic (Lemmers et al. 2010a). Moreover, there is a homologous qA-type D4Z4 array on Chromosome 10q that shares 98% identity with the 4q D4Z4 array (Bakker et al. 1995; Cacurri et al. 1998; Lemmers et al. 2002), yet 10qA alleles contain a single-nucleotide variant (SNV) within the PAS that renders them nonpathogenic (Lemmers et al. 2010a, 2022). Further D4Z4 subhaplotypes have been identified based on other genetic variants within and adjacent to the D4Z4 array (Lemmers et al. 2010b), including two variants of the 4qA allele (4qAS and 4qAL) that differ in the lengths of their distal D4Z4 units, but have been found to be equally pathogenic (Lemmers et al. 2018b). These factors should be accounted for to provide accurate diagnoses for FSHD.
Although previous studies have done much to advance our understanding of FSHD, many aspects of D4Z4 genetics and epigenetics remain poorly understood. It is still unclear the exact mechanisms by which epigenetic factors normally interact with D4Z4 repeats to mediate silencing of the array. Moreover, even amongst patients with the same number of D4Z4 repeats, there is great variability in onset, severity and penetrance of FSHD (Goto et al. 2004; Salort-Campana et al. 2015; Mul et al. 2018b; Ruggiero et al. 2020), suggesting the existence of as-yet unresolved modifiers of the disease. A prominent example of this is the “gray zone” in the repeat number threshold for FSHD1, where 4qA alleles with 8–10 repeat units can give rise to FSHD, but are also found in unaffected control individuals (Giardina et al. 2024). Additionally, Bosma arhinia microphthalmia syndrome (BAMS), another phenotypically distinct human disease caused by pathogenic SMCHD1 variants, has also been associated with D4Z4 hypomethylation (Shaw et al. 2017), yet most BAMS patients do not have the skeletal muscle manifestations of FSHD (Gordon et al. 2017; Mul et al. 2018a). These outstanding questions all highlight the need for methods that can more comprehensively characterize the genetics and epigenetics of D4Z4 regulation.
Until recently, study of D4Z4 has been limited by the low resolution and/or low read length of prior techniques. Southern blotting has long been the “gold standard” for FSHD molecular testing, and uses two separate sets of restriction enzyme digestions and probe hybridizations to determine (a) the number of D4Z4 units in 4q alleles (distinguished from 10q alleles by haplotype-specific SNVs that create 4q-specific XapI and 10q-specific BlnI restriction sites), and (b) the A/B haplotype. Minimally, a typical diagnostic pipeline for FSHD, as outlined in the latest guideline recommendations (Giardina et al. 2024), may involve clinical assessment followed by a Southern blot assay to count the D4Z4 repeat number of 4q alleles. If this first assay shows that the patient does not have a 4q allele of 1–7 repeat units (strongly suggestive of FSHD1), additional assays may be required to determine A/B haplotype (additional Southern blotting assay), evaluate FSHD2-associated genes (exome sequencing), or assess for D4Z4 hypomethylation (bisulfite sequencing). Using this diagnostic approach, it can take months to years for a patient to receive a definitive FSHD diagnosis. Moreover, more complex cases involving mosaicism and complex D4Z4 alleles, including rare 4qA haplotypes, D4Z4 duplications, hybrid arrays, D4Z4 proximal extended deletion (DPED) alleles, and 4q/10q translocations, can give rise to false positives or false negatives, or are often unable to be resolved (Giardina et al. 2024).
Newer technologies such as molecular combing and optical genome mapping have enabled more in-depth characterization of complex D4Z4 alleles that are missed by traditional technologies (Nguyen et al. 2011; Vasale et al. 2015; Nguyen et al. 2017), and are recognised as potential first-line alternatives to Southern blotting for repeat number counting and A/B haplotyping (Giardina et al. 2024). Nevertheless, these assays are still limited by their inability to resolve the base-level D4Z4 sequence, and do not provide methylation information. Several groups have also developed methylation assays that have been shown to accurately diagnose FSHD1 and FSHD2. These methods include a Southern blot–based assay that assesses methylation at the CpG methylation-sensitive FseI restriction enzyme site in the proximal D4Z4 repeat (van Overveld et al. 2003; Lemmers et al. 2015); a methylated DNA immunoprecipitation (MeDIP) method targeting three regions (5′, mid, 3′) within the D4Z4 repeat (Gaillard et al. 2014); and more recent bisulfite-sequencing-based assays that target the DUX4 5’/DR1 region (Hartweck et al. 2013; Jones et al. 2014), the final D4Z4 repeat and the region immediately distal to it for 4qA-/PAS-positive alleles (Jones et al. 2014; Calandra et al. 2016), or highly informative individual CpG sites within the DUX4-PAS and DR1 region (Caputo et al. 2022a; Strafella et al. 2024). However, these assays either average the methylation signal of specific D4Z4 subregions/CpGs across all 4q and 10q repeat units, or specifically target the proximal or distal regions of the array, limiting the study of array-wide D4Z4 methylation patterns.
More recently, several studies have shown the potential of long-read Oxford Nanopore Technologies (ONT) sequencing for the study of the D4Z4 macrosatellite array (Hiramuki et al. 2022; Butterfield et al. 2023; Lemmers et al. 2023; Yeetong et al. 2023; Huang et al. 2024; Li et al. 2024; Wang et al. 2024). Given ONT sequencing is able to simultaneously reveal the genetic sequence and methylation status of the D4Z4 region, the recent guideline updates (Giardina et al. 2024) have identified it as a promising option for future FSHD diagnostics. In this study, we set out to develop an all-in-one, long-read-based workflow for the genetic and epigenetic analysis of FSHD (D4Z4End2End), combining targeted and whole-genome ultra-long sequencing to capture full-length 4q and 10q D4Z4 arrays as well as a panel of FSHD2-associated genes, followed by read-level annotation and haplotyping. We apply D4Z4End2End to the study of several FSHD1, FSHD2, BAMS, and control samples to assess its ability to resolve the genetics and methylation of complete sets of patient 4q and 10q alleles.
Results
Haplotyping and annotation of 4q/10q long-read sequencing data to characterize D4Z4 alleles
We extend upon previous studies (Supplemental Table S1) by developing an all-in-one, long-read-based workflow for the genetic and epigenetic analysis of FSHD: D4Z4End2End. D4Z4End2End was designed to process and visualis long-read sequencing data covering the 4q35 and 10q26 regions, including assignment to Chromosome 4q/10q, determination of A/B haplotype, counting of D4Z4 repeat units, and highlighting of reads that span the entire D4Z4 array (Fig. 1B). Reads that overlap the D4Z4 array were identified by aligning the raw reads against the sequence of a single D4Z4 repeat unit. This avoids potential biases of using a specific genome reference. Alignment of D4Z4-containing reads to the 4q and 10q reference sequences from CHM13v2.0 (containing 4qA and 10qA, Nurk et al. 2022) assigns reads to their chromosome of origin. In addition, XapI and BlnI restriction sites, used to identify each chromosome in current diagnostic approaches, were also assessed within the raw reads, to confirm correct assignment to 4q or 10q. We further assigned the reads to A and B haplotypes based on the presence of pLAM (qA-specific sequence) and qB-specific sequences, respectively. Reads that contained both p13E-11 (a region flanking the proximal end of the D4Z4 array) and either pLAM or qB-specific sequence were classified as spanning reads, and these spanning reads were used to determine the haplotypes and number of D4Z4 units for each allele. To aid in visualization of the results, we created an interactive tool that can be used to explore individual raw 4q and 10q reads that have been processed by the pipeline and annotated with their features (p13E-11, D4Z4 units, pLAM, qB-specific sequence, and XapI and BlnI restriction sites) and haplotypes (4q/10q and A/B) (Figs. 1B, 2).
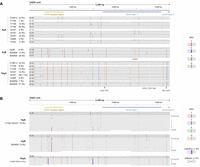
An interactive tool for exploring annotated and haplotyped D4Z4 raw reads from long-read sequencing data. (A) Sequencing data obtained for the 4q35 and 10q26 D4Z4 regions using ultra-long whole-genome sequencing (WGS) and Cas9-targeted sequencing, for sample 17,706 (FSHD1). (Left) Alignment of sequencing data for the D4Z4 region against a single reference genome, such as CHM13v2.0 (32 repeat units for both the 4q35 and 10q26 D4Z4 arrays), leads to large gaps in alignment and potential mis-mapping of reads, making interpretation difficult. (Right) Our pipeline processes the sequencing data to annotate raw reads with their D4Z4 features, enabling accurate repeat unit counting and assignment to 4q/10q and A/B haplotypes. (Top) WGS generates a high total sequencing output (44.86 Gb for sample 17,706) but yields low coverage of the 4q35 and 10q26 regions, which can lead to pathogenic alleles being missed. (Bottom) Cas9-targeted sequencing leads to much higher coverage of the 4q35 and 10q26 regions with a much lower total sequencing output (2.31 Gb for sample 17,706), which enables the capture of low-frequency pathogenic alleles, genetic mosaicism, long full-length arrays, and high numbers of spanning reads. (B) The pipeline is accompanied by an interactive visualization tool that enables exploration of the structure of raw D4Z4 reads, revealing D4Z4 variants such as (left) 4qAS, 4qAL and a newly identified 4qAM allele, as well as (right) 10qB alleles from 4q/10q translocations, and in-cis triplication alleles.
We first used the pipeline to analyze publicly available raw ONT data and assess assemblies published by the Human Pangenome Reference Consortium (HPRC) (Liao et al. 2023), which have previously been studied in the context of FSHD (Salsi et al. 2023). The pipeline was able to successfully identify and annotate raw reads that spanned complete D4Z4 arrays, enabling accurate D4Z4 counting and resolution of 4q and 10q alleles (Supplemental Table S2). Despite the presence of raw spanning reads in the ONT data, assessment of the associated HPRC assemblies showed many of the D4Z4 arrays to be incomplete or split across multiple scaffolds (Supplemental Table S2), creating difficulties for accurate D4Z4 counting and haplotyping. Thus, based on evidence from the raw reads, we find different D4Z4 repeat numbers to those reported previously (Salsi et al. 2023). Additionally, we used the pipeline to process the publicly available raw ONT data used for the CHM13v2.0 assembly (Nurk et al. 2022), and found the number of repeat units in CHM13v2.0’s assembly of the 10q array (33 full 3.3 kb units) to be inconsistent with the raw ONT reads (32 full 3.3 kb units). This shows that there are still outstanding issues in the automated assembly of macrosatellite repeat regions, and demonstrates the need for a tool to visualise the repeat structure of raw reads.
We then used the pipeline to analyze ONT ultra-long whole-genome sequencing (WGS) data from the fibroblasts of two healthy controls, three patients diagnosed with FSHD1, three patients diagnosed with FSHD2, and two patients diagnosed with BAMS (Table 1). The D4Z4 haplotypes of the FSHD1 and FSHD2 patients have previously been studied using Southern blotting and/or molecular combing (Dion et al. 2019; Gaillard et al. 2019; Laberthonnière et al. 2022; Delourme et al. 2023; Gérard et al. 2025), with one of the FSHD1 patients exhibiting somatic mosaicism for a pathogenic 4qA 2RU allele (25% based on Southern blot) and another of the FSHD1 samples harboring a 10qA cis triplication allele (Delourme et al. 2023) (Supplemental Table S3). The D4Z4 haplotypes of the controls and BAMS patients have not previously been studied.
Haplotyping results and number of spanning reads for D4Z4 alleles from FSHD, BAMS, and control fibroblasts, from whole-genome and Cas9-targeted Nanopore sequencing
The pipeline was able to successfully highlight and haplotype 4q and 10q reads from the patient fibroblasts (Table 1), in concordance with the results from Southern blot and molecular combing (Supplemental Table S3). Repeat unit counts based on spanning reads occasionally departed moderately from previous length-based estimates from Southern blot and molecular combing. WGS captured spanning reads for 30 out of 41 alleles, with a maximum of six spanning reads being obtained for a single allele. We captured spanning reads corresponding to the pathogenic, contracted allele for two of the FSHD1 samples, but not for the mosaic FSHD1 sample, perhaps due to the contracted allele having a lower mosaic proportion (25%) (Fig. 2A). The interactive visualization tool also enabled assessment of the length of the distal D4Z4 unit, and we detected both the previously reported 4qAS (∼0.3 kb) and 4qAL (∼1.9 kb) haplotypes. We additionally identified a new, previously unreported 4qA haplotype with a distal D4Z4 unit of length ∼0.6 kb, present in the BAMS1 sample, which we designate “4qAM” (Fig. 2B). All 10qA alleles contained a distal D4Z4 unit of length ∼0.3 kb, similar to 4qAS alleles.
Ultra-long read sequencing also enabled assessment of extended regions of 4q and 10q sequence upstream of and downstream from the D4Z4 array (Fig. 2B). We use the terms “upstream” and “downstream” to refer to genomic regions that are closer to the centromere or telomere relative to the D4Z4 array, respectively. For eight of the 4q alleles, reads were obtained that captured the inverted D4Z4 unit (D4S2463) containing DUX4c that is found ∼42 kb upstream of the 4q D4Z4 array (Wright et al. 1993; Ansseau et al. 2009) (Table 1, Fig. 2B). For three of the samples (12,566, 15,166, and 34,140), 10q alleles were identified that belonged to B haplotypes and contained XapI restriction sites, indicating possible 4q-to-10q translocations upstream of the array. The region of homology between 4q and 10q extends ∼42 kb upstream of the D4Z4 array (Deidda et al. 1995; Delourme et al. 2023), and these alleles were able to be confidently assigned to Chromosome 10q as the ultra-long reads spanned >42 kb of upstream sequence (Fig. 2B). Moreover, the pipeline identified reads that spanned all three arrays of the 10qA in-cis triplication allele of sample FSHD1_3 (37-I1 in Delourme et al. 2023), revealing the allele’s complex structure with nucleotide resolution (Fig. 2B). As has been previously suggested, translocated or duplicated alleles with intact DUX4 and PAS sequences may give rise to DUX4 expression (Lemmers et al. 2018a, 2022), however these complex alleles cannot always be accurately resolved by Southern blotting and molecular combing (Delourme et al. 2023; Giardina et al. 2024). This demonstrates that our pipeline can comprehensively characterize 4q and 10q D4Z4 alleles, including reliable identification of contracted 4qA alleles to confirm the diagnosis of FSHD1, and identification of other structural variants which may contribute to FSHD pathogenesis.
Cas9-targeted sequencing of 4q/10q D4Z4 alleles and FSHD2-associated genes
Although ultra-long WGS is able to capture reads that span the entire 4q and 10q D4Z4 arrays with no prior assumption of the repeat composition or structure, a single PromethION flow cell per patient only provides low coverage. This can lead to pathogenic alleles being missed (Table 1, Fig. 2A), difficulty in creating an accurate consensus sequence of each allele, and in exploring cell-to-cell epigenetic variability or indeed genetic mosaicism.
To address this, we used Cas9-targeted sequencing to enrich for the 4q and 10q D4Z4 regions, aiming to capture the whole set of alleles in each patient. We designed guide RNAs targeting regions upstream of and downstream from the D4Z4 array, for both A and B alleles (Supplemental Table S4). Additionally, we complemented this with guide RNAs designed to target a panel of FSHD2-associated genes (SMCHD1, DNMT3B, and LRIF1) (Supplemental Table S4), with a view to developing an all-in-one genetic and epigenetic research and diagnostic tool for FSHD. This enabled us to obtain high-read-depth sequencing of both Chr 4q35 and Chr 10q26 (Table 1, Fig. 2A) and of SMCHD1, DNMT3B, and LRIF1 exons, respectively (Supplemental Fig. S1).
For all except one of the samples assayed using Cas9-targeted sequencing (three FSHD1 samples, one FSHD2 sample, and one BAMS sample), we were able to determine the full set of 4q and 10q haplotypes by capturing at least one spanning read from each of the patient’s 4q/10q alleles (Table 1). This included the capture of all three 4q alleles from the two mosaic FSHD1 patients (Table 1, Fig. 2A). Although the proportions of spanning 4q reads for each allele from the mosaic samples did not match the expected mosaic proportions, sample 19,187’s 4qA 2RU allele was captured at a much higher frequency than sample 17,706’s 4qA 1RU allele, perhaps reflecting its higher mosaic proportion (50% vs. 25%). Overall, there were far more spanning reads for shorter arrays than for longer arrays, likely due to the greater potential for fragmentation of long reads during library preparation, and preference for short reads during sequencing. Nevertheless, we were able to capture multiple spanning reads for alleles of up to 40 repeat units (∼142 kb), well beyond the maximum read length for Cas9-targeted sequencing recommended by ONT (30 kb).
Genetic analysis of 4q/10q alleles using high-accuracy consensus sequences
For alleles with high read depth from Cas9-targeted sequencing, we used Racon (Vaser et al. 2017) to create full-length consensus sequences for the D4Z4 array. Although many automated assembly tools struggle to accurately reconstruct repetitive alleles (Supplemental Table S2), our consensus-calling approach (see Methods) was able to correct errors while faithfully retaining the structure of the raw D4Z4 reads, including the number of repeat units and unusual variants such as a truncated internal D4Z4 repeat (Supplemental Fig. S3C). This was able to be readily verified by using our interactive visualization tool (Fig. 2) to compare the repeat structure of the consensus sequences to that of the raw reads.
Using the consensus sequences, we were able to reliably analyze SNVs and small insertions/deletions (indels) from 4qA, 4qB, and 10qA alleles. We identified 4q- and 10q-specific variants that were consistent with those seen in CHM13v2.0, including the two SNVs responsible for the 4q-specific XapI site and 10q-specific BlnI site (Fig. 3A). Additionally, we identified qB-specific variants from the BAMS9 4qB alleles, that were consistent with those found in the 4qB allele from GRCh38 (Fig. 3A). Several haplotype-specific variants were present in the DUX4 open reading frame (ORF), including four silent SNVs, a 4qB-specific A > G variant leading to an I229V substitution, and a 10qA-specific 6-bp deletion leading to A340_S341del. Several variants were also identified in the DUX4 promoter region, but none were found to overlap known promoter elements including the GC box, TACAA box, and initiator sequence (Supplemental Fig. S2).
Array-wide and base-level analysis of D4Z4 genetic variants using consensus sequences from high-coverage long-read sequencing. (A) Integrative Genomics Viewer (IGV) coverage plots for alignments of D4Z4 units from the consensus sequences for patient 4q and 10q alleles, showing 4qA-, 4qB- and 10qA-specific genetic variants consistent with CHM13v2.0 and GRCh38. (B) IGV alignments of D4Z4 units from the consensus sequences for representative 4qA, 4qB and 10qA alleles from samples 17,706 and BAMS3. Within each alignment track, full-length D4Z4 units are arranged from top to bottom in order of proximal to distal position within the array. Indels <4 nt which were not consistent between repeat units were hidden to aid in visualization (Supplemental Fig. S3). Consensus sequences were generated using Racon using spanning reads from high-coverage Cas9-targeted ONT sequencing. SNV, single nucleotide variant.
Comparison of the variants between individual D4Z4 units within each 4q and 10q array showed 4q arrays to be more heterogeneous than 10q arrays (Fig. 3B, Supplemental Fig. S3A–E), consistent with previous observations (Salsi et al. 2023). For both 4qA and 4qB arrays, the proximal repeat unit contained a distinct set of variants compared to most other D4Z4 units in the array, although these variants were occasionally also found in internal repeats (Fig. 3B). Variants in the proximal and internal repeat units have previously been used to define D4Z4 subhaplotypes (Lemmers et al. 2010b), however these have largely been restricted to SNVs affecting restriction enzyme sites. The additional 4qA-, 4qB- and 10qA-specific genetic variants identified here provide further insight into D4Z4 subhaplotypes and the composition of the D4Z4 array.
The composition of the D4Z4 array could also be used to reconstruct full-length haplotypes for alleles without spanning reads, and we demonstrated this for the BAMS1 4qAM allele (Supplemental Fig. S4). We also identified a 4qAM allele from the Cas9-sequencing data for sample 19,187 (FSHD1) (Fig. 4), and we found the full-length BAMS1 4qAM allele to be the same as 19,187’s 4qAM allele (Supplemental Fig. S3C), suggesting a common ancestral origin.
Base-level comparison of distal D4Z4 sequences from 4qAS, 4qAM, 4qAL and 10qA alleles. (Top) Alignment of distal D4Z4 sequences beginning from the proximal KpnI site for the final partial D4Z4 unit, and extending into the proximal portion of the pLAM sequence containing DUX4 exon 3. Sequences are extracted from consensus sequences generated using Racon from high-coverage Cas9-targeted ONT sequencing. Breakpoints for the final partial D4Z4 units (vertical arrows) were determined by alignment to a full-length D4Z4 sequence. The start of pLAM is marked as the first nucleotide following the final partial 4qAS D4Z4 unit. The 4qAM and 4qAL alleles contain an extra length of sequence between the end of their final partial D4Z4 units and the start of pLAM, which corresponds to the end of the 4qAS allele’s final partial D4Z4 unit (shown in orange boxes). Locations of primers used for PCR genotyping of 4qAS/4qAM/4qAL (Supplemental Fig. S6, Supplemental Table S5) are shown above their respective sequences; 4AS-F, 4AL-F, and PAS-R (gray) are from Lemmers et al. (2018b), whereas 4AM-F_565 and 4AM-F_345 (brown) were newly designed using the 4qAM consensus sequence. (Bottom) Schematic comparing the distal sequences of 4qAS, 4qAM (newly identified), 4qAL and 10qA alleles.
The consensus sequences also enabled more detailed assessment of the 4q subhaplotype based on sequences proximal and distal to the D4Z4 array. Alignment of the distal D4Z4 sequences of 4qAS, 4qAL and 4qAM alleles showed all three haplotypes to have identical pLAM sequences, containing an intact PAS (Fig. 4, Supplementl Fig. S5). Using the 4qAM consensus sequence, we designed 4qAM-specific primers (Supplemental Table S5, Fig. 4) and validated the presence of the 4qAM allele in sample 19,187 by PCR (Supplemental Fig. S6). To further assess the potential pathogenicity of 4qAM alleles, we also assessed the simple sequence length polymorphism (SSLP) found ∼3.5 kb upstream of the D4Z4 array that has been used to further delineate permissive and nonpermissive D4Z4 subhaplotypes (Lemmers et al. 2007). We found that the SSLP for the BAMS1 4qAM allele corresponded to the 4A161 subhaplotype, which is permissive for FSHD. This suggests that 4qAM alleles may have the potential to be pathogenic, and may need to be considered in genetic testing.
These findings demonstrate the utility of our approach for analyzing D4Z4 alleles at array-wide, base-level resolution, including the potential to further characterize known D4Z4 subhaplotypes, discover new subhaplotypes, and resolve the composition of D4Z4 arrays.
Detection of pathogenic variants in SMCHD1, LRIF1, and DNMT3B
We next applied a variant calling and variant effect prediction pipeline to the whole-genome and Cas9-targeted ONT sequencing data, to assess whether our assay could obviate the need for separate genetic testing. We focused on a panel of FSHD2-associated genes, SMCHD1, LRIF1, and DNMT3B, part of the gene panel for Cas9-mediated enrichment. SMCHD1 genotyping has previously been performed for the patients included in this study (Supplemental Table S3).
We identified pathogenic SMCHD1 variants for three of the four FSHD2 samples (11,440, 15,166, and 11,491) and both of the BAMS samples (BAMS1, BAMS9) that recapitulated the prior genotyping results (Supplemental Table S3). For one of the patients diagnosed as FSHD2 (34,140), no pathogenic variant in SMCHD1, DNMT3B, or LRIF1 was found. Visual inspection of ultra-long read alignments showed no evidence of structural variants at these loci either (Supplemental Fig. S7). The presence of a synonymous variant in SMCHD1 was confirmed (p.L1031), but with no further evidence of pathogenicity, calling into question the diagnosis as FSHD2. This diagnosis was established in early childhood on the basis of a severe muscular dystrophy phenotype; no follow-up information is available for this patient.
The BAMS variants were heterozygous missense variants located in or adjacent to SMCHD1’s N-terminal ATPase domain (sample BAMS1: p.E136G, sample BAMS9: p.D420V). Conversely, the FSHD2 variants were heterozygous splice donor (sample 11,440: c.2338+4A>G [intron 18]; sample 11,491: c.5547+5G>A [intron 44]) or frameshift (sample 15,166: c.4608_4614dup, p.Ala1539Tyrfs*6 [exon 37]) variants found closer towards the C-terminal end of the protein, predicted to lead to SMCHD1 haploinsufficiency and consistent with an FSHD2 diagnosis.
In one of the FSHD1 samples (12,566), we also identified a previously unreported p.R1887L missense variant in exon 45 of SMCHD1 which was predicted to be pathogenic by AlphaMissense (Cheng et al. 2023), with a pathogenicity score of 0.9642. None of the samples had pathogenic variants in LRIF1 or DNMT3B. Notably, whereas some FSHD2-associated genes had only low coverage with whole-genome sequencing, the read depth was significantly increased with Cas9-targeted sequencing, aiding in detection of heterozygous pathogenic variants.
This confirmed the suitability of a single nanopore sequencing assay for 4q haplotyping, 4q structural characterization and variant detection in an FSHD gene panel.
Allele-specific CpG methylation patterns across the D4Z4 array
Array-size-dependent methylation patterns are seen in FSHD1 and control fibroblasts
To assess the methylation patterns of D4Z4 arrays of different lengths and SMCHD1 variant status, we called CpG methylation for the 4q and 10q reads, and used NanoMethViz (Su et al. 2021, 2025) to plot the methylation of individual molecules as well as the smoothed methylation profile across the complete array for each allele.
For FSHD1 and control samples, an oscillatory pattern of D4Z4 methylation was seen within each repeat unit, with low levels of methylation around the DR1 region, which corresponds to the distal portion of a proposed CTCF binding site (Ottaviani et al. 2009; Hartweck et al. 2013), and high levels of methylation around the DUX4 transcription start site (TSS) (Fig. 5A–C, Supplemental Fig. S8A–E), consistent with previous studies (Huichalaf et al. 2014; Butterfield et al. 2023). These oscillatory patterns were present in all noncontracted alleles, but absent from the hypomethylated, contracted FSHD1 alleles. Autocorrelation of the methylation status of CpGs across the D4Z4 array indicated strong correlation between sites separated by ∼3.3 kbp, corresponding to the size of one D4Z4 unit, as well as between sites separated by ∼180 bp (Supplemental Fig. S9), which likely represents nucleosome positioning (Butterfield et al. 2023; Kerr et al. 2023). The high read depth provided by Cas9-targeted sequencing enabled us to see that for each allele, the overall methylation patterns were highly consistent between individual molecules (Fig. 5), but that there was significant intermolecular heterogeneity in methylation state at the level of individual CpG sites (i.e., a given CpG site can be methylated in one read and unmethylated in another; Supplemental Fig. S10).
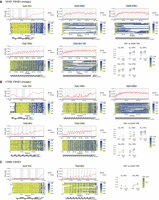
Allele-specific, array-wide D4Z4 methylation profiles for FSHD1 fibroblasts from Cas9-targeted sequencing. Single-molecule and smoothed methylation plots for 4q and 10q D4Z4 alleles from the FSHD1 samples (A) 19,187, (B) 17,706, and (C) 12,566, generated using NanoMethViz (Su et al. 2021, 2025). The locations of DUX4 exons are shaded in gray on the smoothed methylation plot. Annotations for D4Z4 repeat units, CTCF insulator regions, and DUX4 exons are shown below each plot. Mean methylation rates over the DR1 region (positions 563–814 of the D4Z4 KpnI-KpnI unit) (Hartweck et al. 2013) and DUX4 transcription start site (TSS) region (−200 to +200 with respect to the TSS at position 1688 of the D4Z4 KpnI-KpnI unit) within each D4Z4 unit were also plotted for each allele. Points for DR1 and DUX4 TSS regions from the same D4Z4 unit are connected by gray lines.
Differing array-wide methylation patterns were observed for short, intermediate and long D4Z4 arrays (Fig. 5, Supplemental Fig. S8). The short, pathogenic FSHD1 arrays, which ranged from 1 to 3 repeat units, were severely hypomethylated across the full length of the array (Fig. 5A–C and Supplemental Fig. S8C, D, alleles shaded in yellow). Long arrays of >20 repeat units were much more highly methylated, and tended to display a regular, stepwise increase in D4Z4 methylation over the first ten or so repeat units before reaching a constant, high level of methylation (Fig. 5A,B and Supplemental Fig. S8A–E, alleles shaded in blue), similarly to what has been described previously (Butterfield et al. 2023). In contrast, short D4Z4 arrays at the upper end of the pathogenic range for FSHD1 (6–10 repeat units) and intermediate-length arrays (11–20 repeat units) were more variable in their methylation patterns (Fig. 5A–C and Supplemental Fig. S8A–E, alleles shaded in green). Although these arrays were often more highly methylated at their distal ends compared to their proximal ends, this proximal-to-distal increase in methylation did not always occur in a stepwise fashion, as exemplified by the “irregular” methylation patterns seen in several of the alleles from sample 17,706 (Fig. 5B). These observations may partially explain why array sizes in the 1–4 repeat unit range are seen in more clinically severe and early-onset cases of FSHD, whereas longer arrays are associated with variable penetrance and a broad range of clinical severity (Mul et al. 2018b; Goselink et al. 2019).
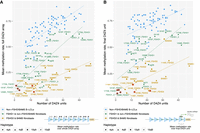
The differences in D4Z4 methylation between short, intermediate and long D4Z4 arrays were also reflected in the global D4Z4 methylation level for each allele, as measured by the proportion of all methylated CpGs across the D4Z4 array (henceforth “global D4Z4 5mC rate).” Overall, we found a positive correlation between the average allelic D4Z4 5mC rate and the number of repeat units for both FSHD1 samples and controls (Pearson correlation coefficient 0.844, Spearman’s correlation coefficient 0.826) (Fig. 6A, Supplemental Fig. S11A). As only the terminal DUX4 gives rise to stable transcripts in permissive 4qA alleles, we additionally assessed the mean methylation of the final full D4Z4 unit, which contains the promoter and exons 1 and 2 of the terminal DUX4. Amongst FSHD1 and control samples, there was a moderate correlation between the number of repeat units and mean methylation rate of the final full D4Z4 unit (“final D4Z4 5mC rate”) (Pearson correlation coefficient 0.701, Spearman’s correlation coefficient 0.732) (Fig. 6B, Supplemental Fig. S11B). As expected, the pathogenic, contracted FSHD1 alleles had very low final D4Z4 5mC rates, ranging from 0.064 to 0.097. However, there were two intermediate-length SMCHD1-wild-type alleles which also had low final D4Z4 5mC rates, the FSHD1_3 4qB 17RU allele (final D4Z4 5mC rate 0.118) and the AG10803 (ag10) 4qB 18RU allele (final D4Z4 5mC rate 0.149). The AG10803 4qB 18RU allele appeared to be hypomethylated across the full length of the array (global D4Z4 5mC rate 0.116), although there were only two spanning reads to support the methylation signal (Supplemental Fig. S8A). These findings once again suggest that in FSHD1 and control fibroblasts, although contracted D4Z4 arrays are consistently hypomethylated, and long D4Z4 arrays are consistently hypermethylated, methylation patterns for intermediate-length arrays are more variable.
Correlation of allelic D4Z4 methylation levels with the number of repeat units. Mean methylation rates of individual alleles across (A) the full 4q/10q D4Z4 arrays or (B) the final D4Z4 unit against the number of D4Z4 units, plotted for alleles from SMCHD1-wild-type fibroblasts (FSHD1 and non-FSHD/BAMS), pathogenic-SMCHD1-variant fibroblasts (FSHD2 and BAMS), and 30 B-lymphoblastoid cell lines (B-LCLs) from the 1000 Genomes Project (Gustafson et al. 2024). Sample 34,140, initially clinically diagnosed as FSHD2, was included in the “non-FSHD/BAMS” group based on the lack of pathogenic SMCHD1, LRIF1, or DNMT3B variants and overall methylation profile. Mean methylation rate was calculated as the total number of methylated CpGs from all reads/the total number of methylated + unmethylated CpGs from all reads, across the defined regions. Regression lines were plotted for each group of samples. Alleles presumed to be the pathogenic alleles for each of the FSHD patients are indicated with red rings. (A) Pearson and Spearman’s correlation coefficients for FSHD1 and non-FSHD/BAMS fibroblasts, FSHD2 and BAMS fibroblasts, and B-LCLs were 0.844 and 0.826, 0.757 and 0.742, and 0.746 and 0.798, respectively. (B) Pearson and Spearman’s correlation coefficients for FSHD1 and non-FSHD fibroblasts, FSHD2 and BAMS fibroblasts, and B-LCLs were 0.701 and 0.732, 0.583 and 0.607, and 0.531 and 0.552, respectively.
B-lymphoblastoid cell lines show similar D4Z4 methylation patterns to FSHD1 and control fibroblasts, but have higher methylation levels
To assess whether these length-dependent methylation patterns are also seen in a larger cohort and other cell types, we analyzed D4Z4 methylation in 30 B-lymphoblastoid cell lines (B-LCLs) from the 1000 Genomes Project (1KGP), using publicly available ONT WGS data from the 1KGP ONT Sequencing Consortium (1KGP-ONT) (Gustafson et al. 2024). We found 83 4q/10q alleles with reads that spanned the whole D4Z4 array, ranging in length from 6 to 43 repeat units (Supplemental Table S6). The patterns of DNA methylation across the array in the B-LCLs were similar to those seen in SMCHD1-wild-type fibroblasts. All arrays had oscillatory patterns of low methylation around the DR1/CTCF insulator region and high methylation around the DUX4 TSS (Supplemental Fig. S12). For long arrays (>20 repeats), methylation levels increased over the first ten or so repeat units then plateaued at a constant, high level across the remainder of the array, or were consistently high across the whole array. Short and intermediate-length arrays (6–20 repeat units) tended to display more regular, stepwise increases in methylation from the proximal to distal end of the array, contrasting with the “atypical” methylation patterns seen in several of the alleles from the FSHD1 and control fibroblasts. Similarly to what we found in fibroblasts, there was a positive correlation between the global D4Z4 5mC and final D4Z4 5mC rates, and the number of D4Z4 units (Fig. 6). However, mean D4Z4 methylation rates were generally higher in the B-LCLs compared to the SMCHD1-wild-type fibroblasts, suggesting that global D4Z4 methylation levels are cell type specific.
Both FSHD2 and BAMS fibroblasts have global D4Z4 hypomethylation
We sought to investigate the effect of pathogenic FSHD2 and BAMS SMCHD1 variants on D4Z4 methylation. In both FSHD2 and BAMS, there was global hypomethylation of D4Z4 arrays compared to similar-length FSHD1 and control samples (Fig. 7A). Despite the differing effects of FSHD2 and BAMS variants on the SMCHD1 protein (predicted haploinsufficiency and missense mutations in the ATPase domain, respectively), FSHD2 and BAMS samples showed similar overall methylation levels and methylation patterns to each other (Fig. 7, Supplemental Fig. S11). Short and intermediate-length arrays were generally severely hypomethylated across the full length of the array. Longer alleles had relatively higher methylation levels, yet were still markedly hypomethylated compared to similar-length SMCHD1-wild-type samples (Fig. 6A), and generally had lower methylation rates for their final D4Z4 units (Fig. 6B) owing to minimal proximal-to-distal increase in array-wide methylation. This was clearly demonstrated by comparison of the 32RU 4qAM alleles from BAMS9 and 19,187_FSHD1 (Figs. 5A, 7B). However, we also identified several reads with very high methylation levels that mapped to the BAMS1 47RU 4qAS allele (Supplemental Fig. S13B), suggesting either the presence of a separate, highly methylated cell population, or an unexpected and sporadic “rescue” of these cells from hypomethylation.
Allele-specific, array-wide D4Z4 methylation profiles for FSHD2 and BAMS fibroblasts. Single-molecule and smoothed methylation plots for 4q and 10q D4Z4 alleles from (A) FSHD2 and (B) BAMS samples, generated using NanoMethViz (Su et al. 2021, 2025). The locations of DUX4 exons are shaded in gray on the smoothed methylation plot. Annotations for D4Z4 repeat units, CTCF insulator regions, and DUX4 exons are shown below each plot. Mean methylation rates over the DR1 region (positions 563–814 of the D4Z4 KpnI-KpnI unit (Hartweck et al. 2013) and DUX4 TSS region (−200 to +200 with respect to the TSS at position 1688 of the D4Z4 KpnI-KpnI unit) within each D4Z4 unit were also plotted for each allele. Points for DR1 and DUX4 TSS regions from the same D4Z4 unit are connected by gray lines. The full-length reference sequences for the BAMS1 4qAM and 4qAS alleles were determined as described in Supplemental Figs. S4 and S13.
In terms of intra- and inter-repeat methylation patterns, oscillations between low methylation around the DR1/CTCF insulator region and high methylation around the DUX4 TSS were variably present in hypomethylated alleles, but were prominent in more highly methylated longer alleles (Fig. 7). Autocorrelation of 5mC signals once again indicated methylation patterns of ∼3.3 kbp and ∼180 bp periodicity (Supplemental Fig. S9), suggesting that the mechanisms that give rise to these patterns are retained in FSHD2 and BAMS.
Complete genetic and epigenetic analysis of in-cis duplicated arrays and upstream inverted D4Z4 units
Although several studies have used molecular combing to study the structure of in-cis duplication alleles (Vasale et al. 2015; Nguyen et al. 2017; Lemmers et al. 2018a; Delourme et al. 2023; Lemmers et al. 2023), this technique is unable to reveal the underlying genetic sequence or epigenetic status, and many aspects of the genetics and epigenetics of these complex alleles have yet to be resolved. Therefore, we sought to use long-read sequencing to further investigate the composition and methylation of in-cis duplicated arrays.
Our pipeline identified in-cis duplications downstream from the main D4Z4 array in two of the FSHD1 alleles (19,187 10qA, FSHD1_3 10qA). Sample 19,187’s complex 10qA allele was composed of a 26RU array followed by a 1RU array, separated by a ∼6.5 kb spacer sequence, with the proximal partial D4Z4 of the duplicated array beginning ∼0.3 kb after the KpnI restriction site (Fig. 8A). Sample FSHD1_3’s complex 10qA allele contained an in-cis triplication, comprising a 15RU array followed by a 2RU array and a 5RU array, separated by two spacer sequences of ∼20 kb, with the proximal partial D4Z4s of the duplicated arrays both beginning ∼1.5 kb after the KpnI restriction site (Fig. 8B). Analysis of the genetic sequence revealed all of the arrays to be 10qA-type, as determined by the presence of BlnI sites and 10qA-type pLAM sequences lacking a functional PAS. Moreover, the spacer sequences and the sequences distal to the duplicated array all corresponded to the sub-telomeric sequence that is normally found immediately distal to the 10qA D4Z4 array.
Analysis of the structure and methylation of duplicated D4Z4 arrays and upstream inverted D4Z4 units. (A), (B), (C) Structure of 4q and 10q alleles identified by the pipeline to have in-cis duplications downstream from the D4Z4 array, alongside smoothed methylation plots showing that these duplicated arrays are hypermethylated. The overall %5mC for each D4Z4 array is shown below the annotation track for the methylation plot. The locations of DUX4 exons are shaded in gray on the smoothed methylation plot. (D) Representative example of the inverted D4S2463 unit located ∼42 kb upstream of the 4q D4Z4 array, which is found to be hypomethylated in FSHD and control fibroblasts and B-LCL samples. (E) The HG00675 cell line from the 1000 Genomes Project (Gustafson et al. 2024) has a 4qB allele with an upstream inverted D4Z4 array of 2.5 repeat units in place of the D4S2463 unit. This upstream inverted array is also hypomethylated.
We also identified the first reported case of a 4qB in-cis duplicated array, present in the B-LCL sample HG00675. The allele contained a proximal array of at least 14RU, followed by an in-cis duplicated array of 5RU. The arrays were separated by a ∼13.7 kb spacer sequence, with the proximal partial D4Z4 of the duplicated array beginning ∼0.8 kb after the KpnI restriction site (Fig. 8C). Both arrays were 4qB-type, and once again, the spacer sequence was identical to the sequence distal to the duplicated array, both corresponding to the 4qB sub-telomeric sequence. These findings mirror those in Lemmers et al. (2023), where the spacer sequence for 4qA-type in-cis duplication alleles was found to correspond to the 4qA sub-telomeric sequence. The shared basic structure of these 4qA, 4qB and 10qA duplication alleles suggests that they emerged via a common underlying mechanism, likely involving intrachromatid gene conversion.
We then assessed the methylation of the duplicated arrays. For all of these alleles, the downstream, contracted arrays were highly methylated, with average methylation levels similar to or higher than those of the noncontracted upstream arrays (Fig. 8A–C). Additionally, for sample FSHD1_3’s in-cis triplication 10qA allele, the average methylation of the proximal 15RU array was higher than might be expected if it were an isolated 15RU array, based on comparison with the global D4Z4 5mC rate of sample FSHD1_3’s 4qB 17RU allele (0.155) and the methylation of intermediate-length arrays in other SMCHD1-wild-type samples (Fig. 6A). Lemmers et al. (2023) found a similar phenomenon for a mosaic 17+9RU 4qA allele and 17+2RU 4qA allele, where the proximal 17RU allele for the 17+9RU array was more highly methylated than for the 17+2RU array. This may indicate the existence of bi-directional methylation spreading mechanisms between proximal and distal arrays, as suggested by Lemmers et al. (2023), or reflect an underlying chromatin state that is dependent upon the additive effect of both duplicated and nonduplicated arrays.
To further characterize the structure and methylation of in-cis D4Z4 repeats, we also assessed the inverted D4Z4 unit (D4S2463) that is found ∼42 kb upstream of the 4q D4Z4 array. Consistent with previous reports (Wright et al. 1993; Ansseau et al. 2009), we found that D4S2463 is a truncated unit of ∼1.5 kb that is homologous to the distal portion of the KpnI-KpnI D4Z4 unit, and contains a homologous DUX4c gene. Contrary to the downstream in-cis duplicated arrays, the upstream inverted repeat was consistently hypomethylated in the FSHD, BAMS, and control fibroblast samples and in the B-LCL samples (Fig. 8D).
Moreover, we identified one B-LCL allele (HG00099 4qB) that had an inverted D4Z4 array of 2.5 repeat units ∼42 kb upstream of the main D4Z4 array, rather than a single D4S2463 unit (Fig. 8E). Several cases of the same allelic variant have also previously been identified using molecular combing (Lemmers et al. 2018a), where the inverted array was suggested to be an expansion of the D4S2463 repeat. Here, we were able to analyze the composition of the inverted array at base-level resolution, and we found that the first inverted D4Z4 unit was ∼1.6 kb and corresponded to the distal ∼1.6 kb of D4Z4, whereas the second and third inverted D4Z4 units were ∼3.3 kb units containing DUX4 and DUX4c sequences, respectively. Once again, Racon (Vaser et al. 2017) was used to create a consensus sequence for the upstream inverted D4Z4 array, and it was found that all three repeat units contained 4qB-type SNVs, whereas the sequence for the last ∼1.5 kb of the final repeat corresponded to the normal sequence for the inverted D4S2463 unit (Supplemental Fig. S14). This suggests that the inverted array may have arisen by insertion of a segment of 4qB array at the breakpoint site for the D4S2463 unit, mediated by partial homology between the D4S2463 unit and the normal D4Z4 sequence; or alternatively, that the 4qB-type inverted array is in fact the ancestral allele, and gave rise to the partial D4S2463 repeat by contraction.
We then assessed the methylation of this inverted array. Similarly to the single inverted D4S2463 units, the inverted array was hypomethylated, with a mean 5mC rate of 0.160. Therefore, overall, we found that downstream, in-cis duplicated arrays were methylated at similar levels to the first D4Z4 array, whereas upstream inverted arrays were hypomethylated similar to the typical D4S2463 unit. This could suggest that methylation spreading mechanisms at the D4Z4 locus, if present, are orientation-dependent, or may provide further support for an insulator mechanism at the D4Z4 locus that separates upstream inverted and downstream noninverted arrays into separate chromatin compartments (Ottaviani et al. 2009; Gaillard et al. 2019).
Discussion
The D4Z4 macrosatellite array is one of the most difficult regions in the genome to study. Traditional techniques such as Southern blotting and short-read sequencing are only able to provide fragmented insights into D4Z4 (epi)genetics, hampering our ability to resolve complex D4Z4 alleles, and to unravel the molecular mechanisms acting at this locus in health and disease. Using long-read nanopore sequencing, here we have developed an optimized protocol and all-in-one analysis workflow for FSHD, D4Z4End2End, that is able to identify full sets of patient 4q and 10q alleles, accurately detect pathogenic variants in FSHD2-associated genes (SMCHD1, DNMT3B and LRIF1), and assess D4Z4 methylation status in a single assay.
The interactive annotation and visualization of raw D4Z4 reads outlined issues with current automated macrosatellite assembly. By annotating and exploring individual raw reads, we were able to more reliably resolve the structure, repeat number, and haplotype of D4Z4 alleles, which is critical for accurate downstream analyses and accurate FSHD diagnoses.
We applied our workflow to the study of primary fibroblast samples from two control individuals, four FSHD1 patients, four FSHD2 patients, and two BAMS patients. Although this represents a smaller cohort compared to several previous ONT studies (Hiramuki et al. 2022; Butterfield et al. 2023; Yeetong et al. 2023; Huang et al. 2024), we were able to attain high coverage and capture longer arrays (up to 42 repeat units (∼140 kb)), cases of mosaicism, and several complex alleles (including entire in-cis duplication and triplication alleles (∼173 kb)), while also providing comparisons between full-length FSHD and BAMS alleles. By generating accurate consensus sequences from high-coverage, full-length patient alleles, we were able to perform detailed analysis of D4Z4 genetic variation, including the identification of haplotype-specific SNVs, and characterization of inter-repeat and inter-array variation. Moreover, we discovered a previously unreported 4qA variant, “4qAM,” which differs from the previously identified 4qAS and 4qAL variants by the length of its distal repeat unit, but has an identical pLAM sequence and intact polyadenylation signal. Further studies could elucidate the prevalence and distribution of 4qAM alleles in the general population, whether they can serve as the causative allele for FSHD, and whether they give rise to different DUX4 transcripts, as is the case for 4qAS and 4qAL alleles (Lemmers et al. 2018b). If 4qAM alleles are indeed permissive for DUX4 expression, this would need to be considered in FSHD genetic testing, including updated conversion tables for calculation of repeat unit size using Southern blotting, and the use of 4qAM-specific primers such as the ones we designed in addition to the 4qAS- and 4qAL-specific primers already used for (bisulfite)-PCR assays (Jones et al. 2014; Lemmers et al. 2018b; Giardina et al. 2024).
Repeat number calculations occasionally differed from previously reported numbers in draft pangenome assemblies and Southern blot/molecular combing analyses. Discrepancies with draft pangenome assemblies can be explained by the current assemblers’ difficulties with macrosatellites. Often those regions still require manual scaffolding and curation (Tørresen et al. 2019). Both the assemblies and our analysis use the same raw data, so the direct annotation of multiple spanning reads via D4Z4End2End is more likely to reflect the truth. Discrepancies with Southern blot and molecular combing can arise from their limited resolution on longer alleles (e.g., 37 vs. 40 RU), the specific probes, scanners and software version used, and the caveats of extrapolating RU numbers from array length rather than analyzing the read structure at base resolution with nanopore sequencing. For example, for patient sample 19,187, our sequencing data showed that one internal repeat of a 10qA allele is truncated (∼1.2 kb deletion), which was counted by D4Z4End2End in the RU count (15 RU), but could account for length-based Southern blot/molecular combing estimating one fewer repeat unit (14 RU). The method of counting may also account for differences of +/−1 units with reported lengths, as we exclude partial repeats at either end of the array in accordance with the updated guidelines. For FSHD1_3, all of our RU measurements are one fewer than the reported counts based on Southern blot/molecular combing, which may have been scored using slightly different criteria.
Accurate haplotyping and the capture of full-length spanning reads also allowed us to analyze the allele-specific and array-wide methylation profiles of FSHD and BAMS D4Z4 alleles, in aggregate and at single-molecule resolution. We were able to observe broad patterns in D4Z4 methylation that correlate with D4Z4 array length and SMCHD1 status, including clear hypomethylation of contracted arrays in FSHD1, and of all 4q and 10q arrays in FSHD2. Notably, one of the samples that had been clinically diagnosed as FSHD2 (34,140) displayed comparatively high levels of methylation for all alleles, consistent with the absence of contracted arrays or pathogenic SMCHD1, DNMT3B, or LRIF1 variants. This illustrates that our workflow can be used to confirm a diagnosis of FSHD in patients with clinical manifestations of the disease, or conversely, to suggest an alternative diagnosis.
High-coverage and full-length methylation profiles also yielded several interesting findings. Many arrays showed a stepwise proximal-to-distal increase in methylation, however we observed some arrays with “atypical” methylation patterns, especially in intermediate-length fibroblast alleles. Of note, overall DNA methylation patterns were highly consistent between individual molecules from the same allele, even for arrays with atypical methylation patterns, although not at the level of individual CpG sites. This aligns with observations from previous studies, which have shown that overall DNA methylation patterns can be stably inherited in clonal populations while being heterogeneous at the per-base level, which may represent differences in nucleosome phasing between molecules, stochasticity in the de novo and maintenance activities of epigenetic modifiers, and the influence of the methylation states of neighboring CpGs (Busto-Moner et al. 2020; Wang et al. 2020; Kerr et al. 2023). Future work should reveal whether those full-length D4Z4 methylation characteristics hold in other relevant cell types, such as myocytes to investigate disease pathogenesis and primary peripheral blood cells which could be used for diagnostic testing.
Previous studies have also indicated that BAMS is associated with D4Z4 hypomethylation in a similar manner to in FSHD2 (Shaw et al. 2017), and here we have performed the first full-length analysis of methylation at the D4Z4 array in BAMS patients. Our results confirm that, at least in primary fibroblasts, D4Z4 methylation patterns are highly similar between BAMS and FSHD2, with retention of oscillatory patterns at the ∼3.3 kbp and ∼180 bp levels, but with overall marked hypomethylation. So far, there has only been one reported case of a patient with overlapping symptoms of BAMS and FSHD, on the background of a FSHD2-permissive D4Z4 genotype (Shaw et al. 2017). Shaw et al. (2017) and Mohassel et al. (2022) identified several other BAMS patients without clinical FSHD who had potentially permissive D4Z4 genotypes, however array size information was not available for all patients, D4Z4 methylation values were aggregate results obtained via bisulfite sequencing, and for young patients FSHD onset might not have been reached (as it typically manifests in young adults). Further genotyping and allele-specific methylation analysis could be used to more comprehensively characterize FSHD-permissive alleles in BAMS patients, and explore the possibility of as-yet undiscovered mechanisms that protect these patients from FSHD.
Finally, we also characterized several complex D4Z4 alleles containing duplicated D4Z4 arrays. Our results suggest that 4qA, 4qB and 10qA duplication alleles likely arise via the same mechanism, which results in in-cis duplications of the distal end of the subtelomere in the noninverted orientation. We found that downstream in-cis duplicated arrays are highly methylated, whereas upstream inverted D4Z4 repeats are hypomethylated, which may suggest orientation-dependent long-range DNA methylation spreading mechanisms, inheritance of methylation patterns from the ancestral array, or insulator mechanisms that generate local differences in chromatin organization. Lemmers et al. (2023) found that in-cis duplication alleles may be able to give rise to FSHD-level DUX4 expression and cause clinical FSHD in the absence of D4Z4 hypomethylation, demonstrating the need to further characterize the epigenetic mechanisms mediating DUX4 repression.
Although D4Z4End2End was developed and tested as a research/diagnostic-enabling pipeline only, and roll out to larger cohorts in a clinical laboratory environment will be crucial, we anticipate that our approach has the potential to greatly simplify and streamline FSHD diagnostics. The field of long-read sequencing diagnostics is still emerging and there are no commonly accepted guidelines yet regarding data acquisition or analysis (Eisfeldt et al. 2025). For FSHD specifically, we propose the following principles as a starting point. For FSHD diagnosis, only the short and demethylated 4qA allele is clinically relevant. In principle, as few as two independent full-length reads spanning this allele could be sufficient for a confident diagnosis, because the large structural elements (pLAM, D4Z4 repeats) and pronounced hypomethylation do not demand base-level precision. Thus as little as 15× coverage with read lengths exceeding 70 kb (longer than the pathogenic threshold for both FSHD1 and 2) should be adequate for most samples. FSHD2 could potentially still be diagnosed with nonspanning reads if key indicators such as low methylation, 4qA haplotype, and a pathogenic SMCHD1 or related variant are evident, therefore 40 kb reads might generally be sufficient. In mosaic cases, the required coverage scales with the variant allele frequency; for example, to detect a mosaic variant at 10% frequency, total coverage of the 4q35 region should increase to approximately 75× (although the enrichment bias for shorter fragments in Cas9-targeted sequencing would decrease this requirement in practice). In addition, 15×–20× coverage is generally sufficient to detect small and large germline variants in FSHD-associated genes (Kolesnikov et al. 2024; Smolka et al. 2024). Live analysis could allow stopping of data acquisition as soon as a confident diagnosis is reached. Although the Cas9 protocol is no longer officially supported by ONT for the current R10.4.1 flow cell chemistry, optimization efforts from other groups ensure that this approach remains viable (Scholz et al. 2024; Benarroch et al. 2025). Several adaptations to the protocol could optimize it even further for use in clinical testing. First, in this study we have sequenced DNA from primary fibroblasts derived from skin biopsies, however for diagnostic testing, peripheral blood samples would be more suitable in terms of decreased cost and invasiveness, and increased acceptability to patients. Previous studies that have performed nanopore sequencing of peripheral blood have also shown reduced D4Z4 methylation of pathogenic alleles compared to controls (Hiramuki et al. 2022; Butterfield et al. 2023) as well as in-cis duplication alleles and somatic mosaicism (Lemmers et al. 2023). Second, in our study we use a single flow cell per patient sample, however we envisage that real-time analysis could facilitate dynamic processing of the sequencing data using the haplotyping pipeline, so that runs can be stopped once enough spanning reads are reached. This could allow multiple samples to be run on a single flow cell after sequential washes and reloads, decreasing sequencing costs. Using one flow cell per patient brings the reagent cost to about USD1500; as batching is not required, turnaround can be very quick, 1–3 days, and the single assay can detect both FSHD type 1 and type 2. By comparison, FSHD testing via Southern blot is around a third of the price but turnaround is typically 6–8 weeks, although batching requirements can draw this out to 6 months; if the result is negative for FSHD type 1, further tests for FSHD type 2 increase the time and cost. Third, adaptive sampling has recently emerged as an alternative to Cas9 for targeted sequencing (Loose et al. 2016), and may be able to further simplify library preparation while also capturing sequences further upstream of and downstream from the D4Z4 array, obviating the need for separate whole-genome sequencing. Adaptive sampling targets regions of interest based on an easily editable browser extensible data (BED) file; therefore, another benefit of this approach is that it could allow additional genetic loci to be added with the discovery of new causative variants for FSHD, and enable optional inclusion of loci responsible for other neuromuscular diseases (NMDs), with no added cost or complexity in library preparation. Indeed, previous studies have demonstrated the efficacy of adaptive sampling for diagnosing short-tandem repeat (STR) expansion-associated NMDs (Miyatake et al. 2022; Stevanovski et al. 2022), and we imagine that STRs, the D4Z4 region, FSHD2-associated genes, and causative loci for other genetic NMDs could be combined into a single neuromuscular gene panel for targeted sequencing.
Finally, there are still many unknowns surrounding D4Z4 regulation, and our workflow could help to answer fundamental questions in macrosatellite biology, such as further investigating the dynamic changes in D4Z4 methylation across different cell types, stages of development, and disease states. Indeed, in addition to embryonic development and FSHD, DUX4 expression is also seen physiologically in the human testis and thymus, and pathologically in several solid cancers, inflammatory disorders, and herpesvirus infection (Mocciaro et al. 2021). Elucidating the epigenetic changes in these contexts may help to reveal the underlying mechanisms of DUX4 silencing and reactivation, paving the way for the identification of new therapeutic targets. Additionally, with the recent development of simultaneous profiling techniques such as DiMeLo-seq (Altemose et al. 2022), nanoNOMe (Lee et al. 2020) and nanoHiMe-seq (Yue et al. 2022), the long-read, single-molecule approach presented here has the potential to be extended to the exploration of even more dimensions of D4Z4 regulation, such as SMCHD1 and CTCF binding, nucleosome occupancy, and histone modifications, alongside genetic sequence and methylation. Our workflow for investigating macrosatellite (epi)genetic landscapes therefore provides a means to explore alterations in the molecular machinery in functional studies, investigate the effects of emerging gene therapies, and map these dynamic and intriguing repetitive regions of the genome with more depth, detail and precision than previously possible.
Methods
Patient sample collection and cell culture
All participants provided signed informed consent for research and publication at the time of recruitment. The study complied with the Declaration of Helsinki. This study received ethics approval from the Walter Eliza Hall Institute (Ethics ID: 20/16B) and the French Agency of Biomedicine (CRBAP-HM OCP05P01E003; Étude D-2023-FSHD). FSHD and BAMS patients were clinically diagnosed and have previously received clinical genetic testing for D4Z4 array size (FSHD and controls), D4Z4 methylation (FSHD) and SMCHD1 genotype (FSHD, BAMS, and controls), as described in Supplemental Table S3. Controls were previously confirmed to have D4Z4 array size >10 units and to have no pathogenic SMCHD1 variant. Primary fibroblasts from FSHD and BAMS patients and controls were obtained via skin biopsies. Fibroblasts were cultured in DMEM with 4.5 g/L D-glucose, FBS South American, Penicillin-Streptomycin 5000 U/mL, and GlutaMAX (Thermo Fisher Scientific), and incubated at 37°C, 5% CO2/9% air in a humidified incubator.
DNA extraction
DNA was extracted from primary fibroblasts using the Monarch high-molecular weight (HMW) DNA extraction kit for cells and blood as per the protocol (NEB T3050) using 300 rpm agitation, and eluted in elution buffer (EB) from the relevant ONT kit used for library preparation (SQK-ULK001, SQK-ULK114 or SQK-CS9109).
ONT library preparation and sequencing
For ultra-long whole-genome sequencing, libraries were prepared using the ONT ultra-long DNA sequencing kit (either SQK-ULK001 or SQK-ULK114) according to the relevant ONT protocol. For Cas9-targeted sequencing, Cas9 crRNAs targeting 4q/10q, SMCHD1, DNMT3B, and LRIF1 (Supplemental Table S3) were designed using CHOPCHOP (v3; Labun et al. 2019) (GRCh38 or CHM13 T2T v1.1, CRISPR-Cas9, nanopore enrichment, with efficiency score parameter “Doench et al. 2014 – only for NGG PAM”). Cas9 RNP formation and DNA library preparation for Cas9-targeted sequencing were performed using the ONT Cas9 Sequencing Kit (SQK-CS9109) as per the ONT protocol, using the Long Fragment Buffer in the last wash to retain fragments of 3 kb and longer. Libraries were loaded on R9.4.1 PromethION flow cells (FLO-PRO002) if prepared using SQK-ULK001 or SQK-CS9109, and on R10.4.1 (FLO-PRO114M) if prepared using SQK-ULK114 (Supplemental Table S7), and sequenced to exhaustion on a PromethION P24 (72 h).
Basecalling
Reads from FAST5 files were basecalled using the latest Guppy version at the time of sequencing (v6.1.1 or v6.3.4) using the modified basecalling model dna_r9.4.1_450bps_modbases_5mc_cg_sup_prom.cfg. Reads from pod5 files from later sequencing runs were basecalled using the latest Dorado version at the time of sequencing (v0.3.2 or v0.3.3), using model dna_r10.4.1_e8.2_400bps_sup@v4.2.0 with the argument ‐‐modified-bases 5mCG_5hmCG for concurrent modified basecalling.
Haplotyping pipeline
Processing of FASTQ files to identify, annotate and haplotype 4q and 10q reads was performed using a custom pipeline, D4Z4End2End. Briefly, reads were mapped against a D4Z4 reference sequence (the first full KpnI-KpnI D4Z4 unit from the 4q array of CHM13v1.0) using minimap2 (v2.24, Li 2018), to identify D4Z4-containing reads and count the number of D4Z4 units. These reads were then mapped against the 4q and 10q regions of CHM13v2.0, and secondary and supplementary alignments were filtered out using samtools view -F 2304 (Li 2011), to identify 4q- and 10q-specific D4Z4 reads and assign them to 4q or 10q haplotypes. D4Z4-containing reads were also mapped against reference sequences for p13E-11 (from Hewitt et al. 1994), pLAM (sequence from end of last D4Z4 to end of DUX4 exon 3 in 4q allele of CHM13v2.0), and the 4qB distal region (from GRCh38) for read annotation. Reads containing pLAM and qB-sequence were assigned to haplotype A and B, respectively. Reads containing both p13E-11 and a D4Z4-distal feature (pLAM or qB-sequence) were classified as spanning reads, and all other reads were classified as nonspanning reads. Reads were also searched for exact matches for XapI sites, BlnI sites, and 4qA/10qA-specific poly(A)-signal sequences using regular expression matching in Python. Noise-Cancelling Repeat Finder (v1.01.00, Harris et al. 2019) was used to search reads for beta-satellite repeats (from Greig and Willard 1992) and telomeric repeats. The interactive visualization tool for exploration of haplotyped reads was created using D3.js and is publicly available alongside the D4Z4End2End workflow. The haplotyping pipeline was used to process the ultra-long whole-genome sequencing and Cas9-targeted sequencing data from all patient samples, and publicly available ONT sequencing data for CHM13v2.0 from the Telomere to Telomere (T2T) Consortium (accessed from s3://human-pangenomics/T2T/CHM13/nanopore/rel8-guppy-5.0.7/reads.fastq.gz), 1KGP-ONT (Gustafson et al. 2024) data available on https://s3.amazonaws.com/1000g-ont/index.html, and Human Pangenome Consortium draft assemblies and sequence data available at GitHub (https://github.com/human-pangenomics/HPP_Year1_Assemblies) (Liao et al. 2023).
D4Z4 genetic analysis
For comparison of the haplotyping pipeline results for CHM13 and the HPRC cell lines to their corresponding assemblies (CHM13v2.0 and HPRC assemblies), D4Z4 arrays in the assemblies were annotated by mapping against the D4Z4, p13E-11, pLAM and 4qB reference sequences described above, using minimap2 (Li 2018).
Consensus sequences for patient 4q and 10q alleles were generated from spanning reads identified by the haplotyping pipeline, using a two-step process: (1) minimap2 (Li 2018) for overlap detection, followed by (2) Racon (v1.4.20) (Vaser et al. 2017) for consensus calling, using the ‐‐no-trimming argument and using the longest spanning read as the target sequence for correction. minimap2 (Li 2018) was used to extract 4q/10q D4Z4 units from the consensus sequences and from the CHM13v2.0 and GRCh38 assemblies (GRCh38 accessed from http://hgdownload.soe.ucsc.edu/goldenPath/hg38/chromosomes/), and to map them against a 4qA-type D4Z4 reference sequence. The 4qA-type D4Z4 reference sequence was generated by performing a MUSCLE (v3.8) (Edgar 2004) multiple sequence alignment of all full-length D4Z4 units from the CHM13v2.0 4q allele, and taking the most frequent value at each position (nucleotide or gap). D4Z4 alignments were visualized using Integrative Genomics Viewer (v2.17.4) (Robinson et al. 2011). MUSCLE was also used for multiple sequence alignments of distal D4Z4 sequences, which were visualized using the R package ggmsa (Zhou et al. 2022).
For the BAMS1 sample, WhatsHap (v2.1) (Martin et al. 2016) was used to phase the nonspanning 4qA reads that overlapped the proximal end of the D4Z4 array.
FSHD2 gene panel variant calling
BAM files for patient samples were run through DeepVariant (v1.5.0) (Poplin et al. 2018) using model type ONT_R104, to generate variant call format (VCF) files targeting SMCHD1, DNMT3B, and LRIF1. VCF files were filtered for “PASS” calls using BCFtools (Danecek et al. 2021). VCF files were input into Variant Effect Predictor (VEP) (v107.0) (McLaren et al. 2016) using the T2T-CHM13v2.0 VEP cache file, to annotate variant calls for potential functional impact. Variant calls overlapping SMCHD1, DNMT3B, or LRIF1 with IMPACT=HIGH or IMPACT=MODERATE as output by VEP were further assessed by comparison to previously reported pathogenic SMCHD1 variants (https://databases.lovd.nl/shared/genes/SMCHD1) and variants in healthy controls, as well as variant pathogenicity predictions from AlphaMissense (Cheng et al. 2023).
Methylation analysis
To generate allele-specific methylation plots, haplotyped reads were extracted from the modBAM files and re-aligned using minimap2 to either the allele’s Racon consensus sequence, or a raw read from the allele if no consensus sequence was generated. For samples with high coverage Cas9-targeted sequencing (17,706, 19,187, BAMS9), all 4q and 10q reads were mapped against the full set of consensus sequences for the sample’s alleles, to haplotype both spanning and nonspanning reads. For samples with lower coverage, reads included for methylation analysis were either spanning reads, or reads that could be confidently assigned to an allele based on allele-specific differences in SNVs (XapI or BlnI sites for 4q vs. 10q), distal sequences (AS/AM/AL/B haplotype) and/or number of repeat units. Allele-specific methylation plots were generated using NanoMethViz (Su et al. 2021, 2025), using the plot_region() function with smoothing_window=2000. Annotations shown below the methylation plots were generated using minimap2, as described above.
To perform allele-specific autocorrelation of 5mC signals, bedmethyl files were first produced from the re-aligned modBAM files using modkit (v0.2.5) (https://github.com/nanoporetech/modkit) with the ‐‐cpg and ‐‐combine-strands arguments. A vector of length n where n is the length in nucleotides of the reference sequence for the allele was populated with the %5mC (“fraction modified” from modkit) for all CpG sites with coverage ≥5, and “NA” otherwise. The acf() function within R was then used to calculate the autocorrelation between %5mC values across the length of the vector, and the results were plotted for lag values (corresponding to distance in nucleotides between CpG sites) of up to 10,000 nucleotides (approximately three full D4Z4 units) and up to 500 nucleotides. Allele-specific %5mC plots were generated from the bedmethyl files, once again filtering for CpG sites with coverage ≥5, using ggplot2 (v3.5.1) (Wickham 2011). Smoothed lines were generated using the geom_smooth() function using the LOESS method with span = 0.2.
Mean methylation rate was calculated using the region_methy_stats() function within NanoMethViz (Su et al. 2021, 2025), using a threshold of 0.5 to call methylated and unmethylated sites. Regression lines for the scatterplots of mean methylation prevalence versus number of repeats were produced using geom_line(stat=“smooth”, method = “lm”), and Pearson and Spearman’s correlations were calculated using the cor() function within R. For comparison of the mean methylation prevalence of the DR1 site and DUX4 TSSs within each repeat unit, the DR1 site was defined as nucleotides 563–814 of the D4Z4 unit with respect to the start of the KpnI site (Hartweck et al. 2013), and the DUX4 TSS region was defined as −200nt to +200nt with respect to the DUX4 TSS (nucleotide 10,732 in GenBank AF117653.3).
PCR genotyping of the distal 4qAM variant
We designed two forward primers for the novel 4qAM variant, to be compatible with the reverse primer from (Lemmers et al. 2018b) and resulting in 345 and 565 bp amplicons (sequences in Supplemental Table S5). 12.5 μL PCRs were set up for 4qAS, 4qAM and 4qAL alleles, each with 20 ng genomic DNA, 2× GoTaq Green MasterMix (Promega) and 0.25 μM concentration for each primer pair. Following (Lemmers et al. 2018b), the 4qAS amplification used a touchdown protocol whereas the two 4qAM and the 4qAL amplifications used a regular PCR protocol. Touchdown protocol: initial denaturation of 2′ at 95°C, followed by 10 cycles of touchdown PCR: 95°C 30″, 70 > 60°C 30″, 72°C 30″; then 26 cycles (95°C 30″, 60°C 30″, 72°C 30″), and a 5′ final extension at 72°C. Regular protocol: 95°C for 2′, 35 cycles of (95°C 30″, 60°C 30″, 72°C 30″), and a 5′ final extension at 72°C. PCR products were run on a 1.8% agarose gel.
Code availability
The D4Z4End2End pipeline inclusive of the interactive visualization tool is freely available at GitHub (https://github.com/lucindaxiao/D4Z4End2End) under an MIT licence, and archived as Supplemental Code.
Data access
All patient sequencing data generated in this study have been submitted to the European Genome-Phenome Archive (EGA; https://ega-archive.org/) under accession number EGAD50000001551.
Competing interest statement
Q.G. and L.C.X. received travel support from ONT to attend a conference.
Acknowledgments
We thank Stephen Wilcox and Sarah MacRaild from the WEHI Genomics Platform for their assistance with nanopore sequencing; and Kelsey Breslin and Hannah Vanyai for their help with cell culture. M.E.R., M.E.B., and Q.G. are supported by Australian National Health and Medical Research Council (NHMRC) Investigator Grants (GNT2017257, GNT1194345 plus GNT2041117, and GNT2007996, respectively). This work was supported by an early career research grant from the Brockhoff foundation and the Marian and E.H. Flack Trust to Q.G. The Walter and Eliza Hall Institute receives support from the Victorian State Government through its Operational Infrastructure Support Program. Additional support was provided by the Australian Government through the National Collaborative Research Infrastructure Strategy (NCRIS) program and an Australian National Health and Medical Research Council Independent Research Institutes Infrastructure Support Scheme (IRIISS) grant (9000719).
Author contributions: Study design: F.M., M.E.B., and Q.G.; data collection: L.C.X., B.S.J., J.L., F.M., and Q.G.; analysis pipeline development: L.C.X., A.S., K.Z., and S.S.; analysis and interpretation of the results: L.C.X., A.S., S.X., B.R., M.E.R., F.M., M.E.B., and Q.G.; supervision: M.E.R., M.E.B., and Q.G.; draft manuscript preparation: L.C.X., M.E.R., M.E.B., and Q.G.
Footnotes
-
[Supplemental material is available for this article.]
-
Article published online before print. Article, supplemental material, and publication date are at https://www.genome.org/cgi/doi/10.1101/gr.280907.125.
-
Freely available online through the Genome Research Open Access option.
- Received May 7, 2025.
- Accepted January 1, 2026.
This article, published in Genome Research, is available under a Creative Commons License (Attribution 4.0 International), as described at http://creativecommons.org/licenses/by/4.0/.