Systematic detection of brain protein-coding genes under positive selection during primate evolution and their roles in cognition
Abstract
The human brain differs from that of other primates, but the genetic basis of these differences remains unclear. We investigated the evolutionary pressures acting on almost all human protein-coding genes (N = 11,667; 1:1 orthologs in primates) based on their divergence from those of early hominins, such as Neanderthals, and non-human primates. We confirm that genes encoding brain-related proteins are among the most strongly conserved protein-coding genes in the human genome. Combining our evolutionary pressure metrics for the protein-coding genome with recent data sets, we found that this conservation applied to genes functionally associated with the synapse and expressed in brain structures such as the prefrontal cortex and the cerebellum. Conversely, several genes presenting signatures commonly associated with positive selection appear as causing brain diseases or conditions, such as micro/macrocephaly, Joubert syndrome, dyslexia, and autism. Among those, a number of DNA damage response genes associated with microcephaly in humans such as BRCA1, NHEJ1, TOP3A, and RNF168 show strong signs of positive selection and might have played a role in human brain size expansion during primate evolution. We also showed that cerebellum granule neurons express a set of genes also presenting signatures of positive selection and that may have contributed to the emergence of fine motor skills and social cognition in humans. This resource is available online and can be used to estimate evolutionary constraints acting on a set of genes and to explore their relative contributions to human traits.
Modern humans (Homo sapiens) can perform complex cognitive tasks well and communicate with their peers (Dunbar and Shultz 2017). Anatomic differences between the brains of humans and other primates are well documented (e.g., cortex size, prefrontal white matter thickness, lateralization), but how the human brain evolved remains a matter of debate (Varki et al. 2008). A recent study of endocranial casts of Homo sapiens fossils indicates that brain size in early Homo sapiens, 300,000 yr ago, was already within the range of that in present-day humans (Neubauer et al. 2018). However, brain shape evolved more gradually within the Homo sapiens lineage, reaching its current form between about 100,000 and 35,000 yr ago. It has also been suggested that the enlargement of the prefrontal cortex relative to the motor cortex in humans is mirrored in the cerebellum by an enlargement of the regions of the cerebellum connected to the prefrontal cortex (Balsters et al. 2010). These anatomic processes of tandem evolution in the brain paralleled the emergence of motor and cognitive abilities, such as bipedalism, planning, language, and social awareness, which are mainly well developed in humans.
Genetic differences in primates undoubtedly contributed to these brain and cognitive differences, but the genes or variants involved remain largely unknown. Indeed, demonstrating that a genetic variant is adaptive requires strong evidence at both the genetic and functional levels. Only a few genes have been shown to be human-specific. They include SRGAP2C (Charrier et al. 2012), ARHGAP11B (Florio et al. 2015), and NOTCH2NLA (Suzuki et al. 2018), which emerged through recent gene duplication in the Homo lineage (Dennis et al. 2017). The expression of these human-specific genes in the mouse brain expands cortical neurogenesis (Dennis et al. 2012; Florio et al. 2015; Nuttle et al. 2016; Suzuki et al. 2018). Several genes involved in brain function display accelerated coding region evolution in humans. For example, FOXP2 has been associated with verbal apraxia and ASPM with microcephaly (Enard et al. 2002; Montgomery et al. 2014). Functional studies have also shown that mice carrying a “humanized” version of FOXP2 display qualitative changes in ultrasonic vocalization (Enard et al. 2009). However, these reports targeting only specific genes sometimes provide contradictory results (Atkinson et al. 2018). Other studies have reported sequence conservation to be stronger in the protein-coding genes of the brain than in those of other tissues (Miyata et al. 1994; Wang et al. 2006; Tuller et al. 2008), suggesting that the primary substrate of evolution in the brain is regulatory changes in gene expression (King and Wilson 1975; Pollard et al. 2006; Changeux 2017) and splicing (Calarco et al. 2007). In addition, several recent studies have explored the genes subjected to the highest degrees of constraint during primate evolution or in human populations to improve estimations of the pathogenicity of variants identified in patients with genetic disorders (Sundaram et al. 2018; Havrilla et al. 2019). In contrast, fewer studies have systematically detected genes that diverged during primate evolution (Dorus et al. 2004; Nielsen et al. 2005; Huang et al. 2013).
We describe here an exhaustive screening of all protein-coding genes for conservation and divergence from the common primate ancestor, making use of rich data sets of brain single-cell transcriptomics, proteomics, and imaging to investigate the relationships between these genes and brain structure, function, and diseases.
Results
Strong conservation of brain protein-coding genes
We first compared the sequences of modern humans, archaic humans, and other primates to those of their common primate ancestor (inferred from the Compara six-way primate Enredo, Pecan, Ortheus multiple alignments) (Paten et al. 2008) to extract a measurement of evolution for 11,667 of the 1:1 orthologs across primates, selected from the 17,808 protein-coding genes in the modern human genome (available online from https://genevo.pasteur.fr/) (Fig. 1A; see also Supplemental Figs. S1, S2; Kapheim et al. 2015). Our measurement is derived from one of the most widely used and reliable measurements of evolutionary pressure on protein-coding regions, the dN/dS ratio (Yang and Bielawski 2000), also called ω. This measurement compares the rates of nonsynonymous and synonymous mutations of coding sequences. More nonsynonymous mutations than expected signifies positive selection, and fewer signifies selective constraint. We first estimated dN and dS for all 1:1 orthologous genes, because the evolutionary constraints on duplicated genes are relaxed (only the Y Chromosome was excluded from these analyses) (O'Toole et al. 2018). We then adjusted the dN/dS ratio for biases induced by variations of mutation rate with the GC content of codons. Finally, we renormalized the values obtained for each taxon across the whole genome. The final ωGC12 obtained took the form of Z-score corrected for GC content that quantified the unbiased divergence of genes relative to the ancestral primate genome (Kapheim et al. 2015). High positive ωGC12 indicates a genetic signature commonly, but not exclusively, associated with positive evolutionary selection; on the contrary, negative ωGC12 reflects selective constraint.
Evolution of protein-coding genes across tissues and biological functions. (A) Analysis pipeline for the extraction of ωGC12, a corrected and normalized measurement of the evolution of protein-coding genes that behaves like a Z-score and takes into account the GC content of codons. (B) Hierarchical clustering, based on ωGC12, across all protein-coding genes (1:1 orthologs in hominins with medium coverage) (Supplemental Table S1). (C) Gene Ontology (GO) enrichments for the red and blue clusters in B (for all GO terms, see Supplemental Table S2). Horizontal lines indicate 95% confidence intervals. (D) Funnel plot summarizing the evolution of protein-coding genes specifically expressed in different tissues of the human body (Supplemental Table S3). Horizontal and vertical axes indicate, respectively, the effect size and the statistical significance. Circle size indicates the number of proteins in the set. The dashed horizontal line indicates the threshold for significance after Bonferroni correction. Stars indicate the set of genes for which statistical significance was achieved in multiple comparisons after correction, with a bootstrap taking GC12 content and coding sequence length into account. (HS) Homo sapiens; (6-EPO ancestor) the reconstructed ancestral genome of primates based on alignments of Homo sapiens, chimpanzee, gorilla, orangutan, rhesus macaque, and marmoset genomes.
Using the ωGC12 for all protein-coding genes in Homo sapiens, Denisovans, Neanderthals, and Pan troglodytes, we identified two distinct clusters in hominins (Fig. 1B; Supplemental Table S1): one containing “positively selected” genes (PSGs), enriched in olfactory genes (OR = 1.48, P = 8.4 × 10−9), and one with genes under “selective constraint” (SCGs), enriched in brain-related biological functions (Fig. 1C; Supplemental Table S2). This second cluster revealed particularly strong conservation of genes encoding proteins involved in nervous system development (OR = 1.2, P = 2.4 × 10−9) and synaptic transmission (OR = 1.35, P = 1.7 × 10−8).
We investigated the possible enrichment of specific tissues in PSGs and SCGs by analyzing RNA-seq (Illumina Bodymap2 and GTEx), microarray, and proteomics data sets (Methods). For expression data, despite that virtually no gene is expressed only in one tissue, we calculated a tissue specificity score for each gene by normalizing their profile across tissues (for more details, see Supplemental Fig. S3). The results confirmed a higher degree of conservation for protein-coding genes more specifically expressed in the brain (Wilcoxon rank correlation rc = −0.1, P = 4.1 × 10−12, bootstrap-corrected for gene length and GC content) than for those expressed elsewhere in the body, with the greatest divergence observed for genes expressed in the testis (Wilcoxon rc = 0.3, P = 7.8 × 10−11, bootstrap-corrected for gene length and GC content) (Fig. 1D; for a replication with GTEx data, see also Supplemental Table S3; Supplemental Fig. S4). This conservation of brain protein-coding genes was replicated with two other data sets (MicroArray: Wilcoxon OR = −0.18, P = 1.8 × 10−12; mass spectrometry: Wilcoxon rc = −0.21, P = 1.55 × 10−9; bootstrap-corrected for gene length and GC content).
Conservation of protein-coding genes relating to nervous system substructure and neuronal functions
We then used microarray (Su et al. 2004) and RNA-seq (The GTEx Consortium 2015) data to investigate the evolutionary pressures acting on different regions of the central nervous system. Three central nervous system substructures appeared to have evolved under the highest level of purifying selection at the protein sequence level (ωGC12 < 2): (1) the cerebellum (Wilcoxon rc = −0.29, P = 5.5 × 10−6, Bonferroni-corrected) and the cerebellar peduncle (Wilcoxon rc = −0.11, P = 3.2 × 10−4, bootstrap-corrected for gene length and GC content); (2) the amygdala (Wilcoxon rc = −0.11, P = 4.1 × 10−6, bootstrap-corrected for gene length and GC content); and (3) the prefrontal cortex (Wilcoxon rc = −0.1, P = 5.7 × 10−10, bootstrap-corrected for gene length and GC content) (Fig. 2A; Supplemental Table S3). Indeed, it has been suggested that the prefrontal cortex is one of the most divergent brain structures in human evolution (Schoenemann et al. 2005), this diversity being associated with high-level cognitive function (Frith and Dolan 1996). Only one brain structure was expressing more PSG than expected: the superior cervical ganglion (Wilcoxon rc = 0.22, P = 1 × 10−6, bootstrap-corrected for gene length and GC content). This structure provides sympathetic innervation to many organs and is associated with the archaic functions of the fight-or-flight response. The PSGs expressed in the superior cervical ganglion include CARF, which was found to be specifically divergent in the genus Homo. This gene encodes a calcium-responsive transcription factor that regulates the neuronal activity-dependent expression of BDNF (Tao et al. 2002) and a set of singing-induced genes in the song nuclei of the zebra finch, a songbird capable of vocal learning (Whitney et al. 2014). This gene had a raw dN/dS of 2.44 (seven nonsynonymous vs. one synonymous mutation in Homo sapiens compared to the common primate ancestor) and was found to be one of the PSGs with the higher dN/dS value expressed in the human brain.
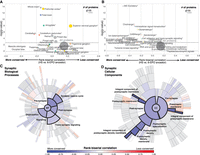
Evolution of brain-related protein-coding genes. (A,B) Funnel plots summarizing the evolution of protein-coding genes specifically expressed in brain substructures (A) and synaptic functions (B); the dashed horizontal line indicates the threshold for significance after Bonferroni correction. Stars indicate sets of genes for which statistical significance was achieved for multiple comparisons with bootstrap correction. (C,D) SynGO sunburst plots showing nested statistically conserved (blue) biological processes (C) and cellular components (D) of the synapse. The circle in the center represents the root node, with the hierarchy moving outward from the center. A segment of the inner circle bears a hierarchical relationship to those segments of the outer circle which lie within the angular sweep of the parent segment.
We then investigated the possible enrichment of PSGs and SCGs in brain-specific Gene Ontology terms. All pathways displayed high overall levels of conservation, but genes encoding proteins involved in glutamatergic and GABAergic neurotransmission were generally more conserved (Wilcoxon rc = −0.25; P = 9.8 × 10−6, Bonferroni-corrected) than those encoding proteins involved in dopamine and peptide neurotransmission and intracellular trafficking (Fig. 2B; Supplemental Table S3). The recently released ontology of the synapse provided by the SynGO consortium (http://syngoportal.org) was incorporated into this analysis, not only confirming the globally strong conservation of the synapse but also revealing its close relationship to trans-synaptic signaling processes (Wilcoxon rc = −0.21, P = 4.5 × 10−5, Bonferroni-corrected) and to postsynaptic (rc = −0.56, P = 6.3 × 10−8, Bonferroni-corrected) and presynaptic membranes (Wilcoxon rc = −0.56, P = 7 × 10−8, Bonferroni-corrected) (Fig. 2C,D).
Positively selected genes and their correlation with brain expression and function
We focused on the genes situated at the extremes of the ωGC12 distribution (>2SD) (Fig. 3A; Supplemental Table S4) and those fixed in the modern Homo sapiens population (neutrality index < 1), to ensure that we analyzed PSGs with signs of strong positive selection. Only 139 of these 352 highly PSGs were brain-related (impoverishment for brain genes, Fisher's exact test OR = 0.66, P = 1 × 10−4), listed as synaptic genes (Ruano et al. 2010; Lips et al. 2012), specifically expressed in the brain (+2SD for specific expression) or related to a brain disease (extracted systematically from Online Mendelian Inheritance in Man [OMIM]: https://www.omim.org; and Human Phenotype Ontology [HPO]: https://hpo.jax.org/app/). For comparison, we also extracted the 427 SCGs under very strong selective constraint, 299 of which were related to the brain categories listed above (enrichment for brain genes, Fisher's exact test OR = 1.26, P = 0.0032).
Brain protein-coding genes and human diseases. (A) Distribution of ωGC12 and Venn diagrams describing SCGs and PSGs situated at the extremes of the ωGC12 distribution (>2SD) specifically expressed in the brain (genes with specificity Z-score > 2 in any brain-related tissues of Figs. 1D and 2A), related to the synapse or brain diseases (Supplemental Table S4). (*) Addition of four genes (FARSB, KRT14, NPHS1, RSPH1) containing Homo sapiens–specific mutations predicted as deleterious (CADD>15). (B) Odds ratios for protein-coding gene sets related to brain diseases (Fisher's exact test; asterisks indicate P-values significant after Bonferroni correction; horizontal lines indicate 95% confidence intervals).
Using these 427 SCGs and 352 PSGs, we first used the Brainspan data available from the specific expression analysis (SEA) to confirm that the population of genes expressed in the cerebellum and the cortex was enriched in SCGs (Supplemental Fig. S5). Despite this conservation, based on the adult Allen Brain Atlas, we identified a cluster of brain subregions (within the hypothalamus, cerebral nuclei, and cerebellum), more specifically expressing PSGs (Supplemental Fig. S6). Analyses of the human cerebral cortex single-cell RNA-seq (Fig. 4A; Supplemental Table S5; Nowakowski et al. 2017) also revealed an excess of PSGs expressed in the choroid plexus (the primary function is to produce cerebrospinal fluid), in the medial ganglionic eminence (MGE-div; implicated in the production of GABAergic interneurons and their migration to the neocortex during development) (Brazel et al. 2003), and the radial glial (RG) cells.
Evolution of protein-coding genes expressed in different cell types. (A–C) Funnel plots summarizing the evolution of protein-coding genes specifically expressed in different cell types within the human cerebral cortex (Supplemental Table S6; Nowakowski et al. 2017) (A), human cortical layers (Supplemental Table S6; Hodge et al. 2019; Tasic et al. 2018) (B), and the mouse cerebellum (Supplemental Table S6; Carter et al. 2018) (C). (D) Venn diagram of the PSGs expressed specifically in those cell types, with the corresponding Protein–Protein Interaction network (STRINGdb) (Jensen et al. 2009) and their annotated association with micro- and macrocephaly (HPO) (Köhler et al. 2019). (EN-V1) primary visual cortex neurons; (RG-early) radial glia early cortical progenitors; (MGE-div) medial ganglionic eminence dividing cells; (Exc) excitatory; (L3-5) layers 3–5; THEMIS, UBE2F, PLA2G7, and so forth are cell type markers.
Using a second RNA-seq data set from the human cortex (Tasic et al. 2018; Hodge et al. 2019), we identified five cell types, all from layer 3 or 5, expressing PSGs more than expected using a stringent Bonferroni and bootstrap correction for gene length and GC content (Fig. 4B; Supplemental Table S5). Among them, two groups of excitatory neurons, THEMIS-PLA2G7 and FEZF2-SCN7A, express several PSGs involved in DNA damage response (Arcas et al. 2014) and mutated in patients with microcephaly such as BRCA1, NHEJ1, RNF168, and TOP3A.
We investigated organoid and human cortex data sets that previously revealed seven clusters of cells (Camp et al. 2015). Overall, the marker genes of these clusters are on average strongly constrained compared to the rest of the genome (Supplemental Fig. S7). Some PSGs are however expressed in these cells, such as CDC25C, FRMD4B, NHSL1, NUSAP1, and PLEKHA5.
In single-cell transcriptomic studies of the mouse cerebellum (Carter et al. 2018), we found that cells expressing cilium marker genes, such as the dynein light chain roadblock-type 2 (DYNLRB2) and the meiosis/spermiogenesis associated 1 (MEIG1), were the principal cells with higher levels of PSG expression (Fig. 4C; Supplemental Table S5). Those “ciliated cells” were not anatomically identified in the cerebellum (Carter et al. 2018), but their associated cilium markers were found to be expressed at the site of the cerebellar granule cells (Lein et al. 2007). These cells may, therefore, be a subtype of granule neurons involved in cerebellar function. The PSGs expressed in these ciliated cells code for the tubulin tyrosine ligase-like family, member 6 (TTLL6), TOP3A, the dynein cytoplasmic 2 light intermediate chain 1 (DYNC2LI1), and LCA5, coding for a component of the axoneme of ciliated cells. Some of these PSGs are also involved in human brain diseases such as microcephaly, macrocephaly, and Joubert syndrome (Fig. 4D, and below).
Finally, we assessed the potential association with brain functions, by extracting 19,244 brain imaging results from 315 fMRI-BOLD studies (T and Z-score maps) (for the complete list, see Supplemental Table S6) from NeuroVault (Gorgolewski et al. 2015) and comparing the spatial patterns observed with the patterns of gene expression in the Allen Brain Atlas (Hawrylycz et al. 2012; Gorgolewski et al. 2014). The correlation between brain activity and PSG expression was stronger in subcortical structures than in the cortex (Wilcoxon rc = 0.14, P = 2.5 × 10−248). The brain activity maps that correlate with the expression pattern of the PSGs (Supplemental Table S7) were enriched in social tasks (empathy, emotion recognition, theory of mind, language; Fisher's exact test P = 2.9 × 10−20, OR = 1.72, CI95% = [1.53, 1.93]). We also observed this enrichment for expression pattern of the SCGs (Fisher's exact test P = 1.2 × 10−12, OR = 1.16, CI95% = [1.11, 1.22]); however, it was significantly less enriched than for the expression pattern of PSGs (Fisher's exact test P = 0.0004, OR = 0.83, CI95% = [0.75, 0.92]).
Positively selected genes and their relationship to brain disorders
Our systematic analysis revealed that SCGs were more associated with brain diseases or traits than PSGs (Fig. 3B), particularly for intellectual disability (P = 8.13 × 10−6, OR = 0.34 CI95% = [0.21, 0.56], Bonferroni-corrected) and autism (P = 0.0005, OR = 0.26, CI95% = [0.11, 0.59], Bonferroni-corrected). We also identified 42 high PSGs associated (based on OMIM and HPO data) with several human diseases or conditions, such as micro/macrocephaly, autism, or dyslexia (Supplemental Table S4).
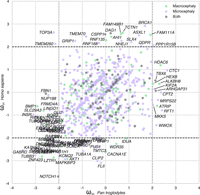
A comparison of humans and chimpanzees with our common primate ancestor revealed several protein-coding genes associated with micro/macrocephaly with different patterns of evolution in humans and chimpanzees (Fig. 5). Some genes displayed a divergence specifically in the hominin lineage (AHI1, ASXL1, BRCA1, CSPP1, DAG1, FAM111A, FAM149B1, GRIP1, NHEJ1, QDPR, RNF135, RNF168, SLX4, TCTN1, TMEM70, TMEM260, and TOP3A) or in the chimpanzee (ALKBH8, ARHGAP31, ATRIP, CPT2, CTC1, HDAC6, HEXB, KIF2A, MKKS, MRPS22, RFT1, TBX6, and WWOX).
Evolution of the protein-coding genes associated with microcephaly or macrocephaly in humans. Scatter plots comparing ωGC12 between Homo sapiens and Pan troglodytes for the microcephaly- and macrocephaly-associated genes.
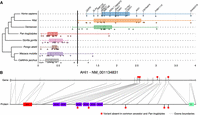
We also identified PSGs associated with communication disorders, such as autism (CNTNAP4, AHI1, FAN1, SNTG2, and GRIP1) and dyslexia (KIAA0319). These genes diverged from the common primate ancestor only in the hominin lineage and were under strong selective constraint in all other taxa (Fig. 6A,B). They all have roles relating to neuronal connectivity (neuronal migration and synaptogenesis) and, within the human brain, were more specifically expressed in the cerebellum, except for GRIP1, which was expressed almost exclusively in the cortex.
Examples of brain disorder–associated protein-coding genes displaying specific divergence in hominins during primate evolution. (A) Representation of 16 genes with dN/dS > 1 in Homo sapiens and archaic hominins but dN/dS < 1 for other primates. (B) Representation of hominin-specific nonsynonymous variants of the AHI1 gene, showing the correspondence with the protein (dotted lines indicate exons); note how two variants lie within the WD40 functional domains. Red stars indicate variants (CADD > 5) relative to the ancestor present in Homo sapiens, Neanderthals, and Denisovans, but not in Pan troglodytes. (WD40) WD40 repeat; (SH3) SRC homology 3; (CC) coiled coils.
The dyslexia susceptibility gene KIAA0319, encoding a protein involved in axon growth inhibition (Paracchini et al. 2006; Franquinho et al. 2017), is one of the PSGs under the strongest positive selection in humans relative to the common primate ancestor (raw dN/dS = 3.9; nine nonsynonymous vs. one synonymous mutations in Homo sapiens compared to the common primate ancestor). The role of KIAA0319 in dyslexia remains a matter of debate, but its rapid evolution in the hominoid lineage warrants further genetic and functional studies.
Finally, several PSGs display very high levels of positive selection in Homo sapiens, but their functions or association with disease remain unknown. For example, the zinc finger protein ZNF491 (raw dN/dS = 4.7; 14 nonsynonymous vs. one synonymous mutation in Homo sapiens compared to the common primate ancestor) is specifically expressed in the cerebellum and is structurally similar to a chromatin remodeling factor, but its biological role remains to be determined. Another example is the CCP110 gene, encoding a centrosomal protein resembling ASPM, but not associated with a disease. Its function suggests that this PSG would be a compelling candidate for involvement in microcephaly in humans. A complete list of the brain SCGs and PSGs is available in Supplemental Table S4 and on the companion website.
Discussion
Positively selected genes and brain size in primates
Several protein-coding genes are thought to have played a significant role in the increase in brain size in humans. Some of these genes, such as ARHGAP11B, SRGAP2C, and NOTCH2NLA (Suzuki et al. 2018), are specific to humans, having recently been duplicated (Dennis and Eichler 2016). Other studies have suggested that a high degree of positive selection in genes involved in micro/macrocephaly may have contributed to the substantial change in brain size during primate evolution (Dorus et al. 2004; Hayward 2004). Several of these genes, such as ASPM (Mekel-Bobrov et al. 2005) and MCPH1 (Evans et al. 2005), seem to have evolved more rapidly in humans. However, the adaptive nature of the evolution of these genes has been called into question (Yu et al. 2007), and neither of these two genes were on the PSG list in our analysis (their raw dN/dS value are below 0.8).
Conversely, our systematic detection approach identified the genes under the strongest positive selection in humans for micro/macrocephaly, the top 10 such genes being AHSG, ASXL1, BRCA1, CSPP1, DAG1, FAM111A, FAM149B1, RNF168, TMEM70, and TOP3A. This list of PSGs associated with micro/macrocephaly in humans can be used to select the best candidate human-specific gene/variants for further genetic and functional analyses, to improve estimates of their contribution to the emergence of anatomic difference between humans and other primates.
As previously shown, our systematic analysis confirms that the major susceptibility gene for breast cancer BRCA1 is under strong positive selection (Lou et al. 2014). BRCA1 is a DNA damage response protein that repairs double-strand breaks in DNA. Heterozygous BRCA1 mutations increase the risk of breast cancer but can also cause neuronal migration defects (Eccles et al. 2005). In sporadic cases, homozygous BRCA1 mutations lead to Fanconi anemia with microcephaly (Mehmet et al. 2016). Several other DNA damage response proteins (Arcas et al. 2014), which are binding partners of BRCA1 (such as SLX4, TOP3A, RNF168, and MCPH1) are also associated with microcephaly. How BRCA1 mutations cause microcephaly in humans remains largely unknown. However, in the mouse, Brca1 mutations strongly reduce the size of the cerebral cortex by affecting the cellular polarity of neural progenitors and preventing the apoptosis of early cortical neuron progenitors (Pulvers and Huttner 2009; Pao et al. 2014). Upper-most cortical layers are not reduced upon Brca1 ablation in mice, and this is consistent with the low levels of apoptosis found in late progenitors and the neurons derived from there. Our analysis of the single-cell RNA-seq data from the human cortex indicates that excitatory neurons from layers 3 and 5 express PSG more than expected, including BRCA1 and several of its binding partners associated with DNA damage response and microcephaly, such as TOP3A, RNF168, and NHEJ1. Further analyses on the role of these genes, which are currently known for their DNA damage response, might shed some light on primate brain evolution.
In addition to brain size, some of the micro/macrocephaly PSGs may have contributed to differences in other morphological features, such as skeleton development. For example, the PSG FAM111A (raw dN/dS = 2.99; seven nonsynonymous vs. one synonymous mutation in Homo sapiens compared to the common primate ancestor) and ASXL1 (raw dN/dS = 1.83; 12 nonsynonymous vs. three synonymous mutations in Homo sapiens compared to the common primate ancestor) are associated with macrocephaly and microcephaly, respectively. Patients with dominant mutations of FAM111A are diagnosed with Kenny-Caffey syndrome (KCS). They display impaired skeletal development, with small dense bones, short stature, primary hypoparathyroidism with hypocalcemia, and a prominent forehead (Unger et al. 2013). FAM111A is a binding partner of BRCA1 and plays a role in DNA damage response, but this protein seems to be also crucial to a pathway governing parathyroid hormone production, calcium homeostasis, and skeletal development and growth. In contrast, patients with dominant mutations of ASXL1 are diagnosed with Bohring-Opitz syndrome, a malformation syndrome characterized by severe intrauterine growth retardation, intellectual disability, trigonocephaly, hirsutism, and flexion of the elbows and wrists with a deviation of the wrists and metacarpophalangeal joints (Hoischen et al. 2011). ASXL1 encodes a chromatin protein required to maintain both the activation and silencing of homeotic genes.
Three genes (AHI1, CSPP1, and TCTN1) in the top 10 of the PSGs associated with human brain diseases, with raw dN/dS > 2, are required for both cortical and cerebellar development in humans. They are also associated with Joubert syndrome, a recessive disease characterized by agenesis of the cerebellar vermis and difficulties coordinating movements. AHI1 is a positive modulator of classical WNT/ciliary signaling. CSPP1 is involved in cell cycle–dependent microtubule organization, and TCTN1 is a regulator of Hedgehog during development.
AHI1 was previously identified as a gene subject to positive selection during the evolution of the human lineage (Ferland et al. 2004; Gould and Walter 2004), but to our knowledge, neither CSPP1 nor TCTN1 has previously been described as diverging during primate evolution. It has been suggested that the accelerated evolution of AHI1 required for ciliogenesis and axonal growth may have played a role in the development of unique motor capabilities, such as bipedalism, in humans (Hayward 2004). Our findings provide further support for the accelerated evolution of a set of genes associated with ciliogenesis.
The possible link between a change in the genetic makeup of the cerebellum and the evolution of human cognition
The emergence of a large cortex was undoubtedly an essential step for human cognition, but other parts of the brain, such as the cerebellum, may also have made significant contributions to both motricity and cognition. In this study, we showed that the protein-coding genes expressed in the cerebellum were among the most conserved in humans. However, we also identified a set of PSGs with relatively strong expression in the cerebellum or for which mutations affected the cerebellar function. As discussed above, several PSGs are associated with Joubert syndrome, including AHI1, CSPP1, and TCTN1, and are essential for cerebellar development. Furthermore, the PSGs expressed in the brain and under the highest positive selection include CNTNAP4, FAN1, SNTG2, and KIAA0319, which also display high levels of expression in the cerebellum and have been associated with communication disorders, such as autism and dyslexia. Finally, the choroid plexus expressed more PSGs than expected and is known to play the role of a paracrine gland to produce the retinoic acid necessary for cerebellum development (Yamamoto et al. 1996).
In humans, the cerebellum is associated with higher cognitive functions, such as visuospatial skills, the planning of complex movements, procedural learning, attention switching, and sensory discrimination (Koziol et al. 2012). It plays a crucial role in temporal processing (Rao et al. 2001) and the anticipation and control of behavior through both implicit and explicit mechanisms (Koziol et al. 2012). A change in the genetic makeup of the cerebellum would, therefore, be expected to have been of great advantage for the emergence of the specific features of human cognition.
Despite this possible link between the cerebellum and the emergence of human cognition, much less attention has been paid to this part of the brain than to the cortex, on which most of the functional studies investigating the role of human-specific genes/variants have focused. For example, SRGAP2C expression is considerably high in the human cerebellum, but the ectopic expression of this gene has been studied in mouse cortex (Charrier et al. 2012; Dennis et al. 2012), in which it triggers human-like neuronal characteristics, such as an increase in dendritic spine length and density. We thus suggest that an exploration of human genes/variants specifically associated with the development and functioning of the cerebellum might shed new light on the evolution of human cognition.
Limitations
The present results have potential limits in their interpretations. Sources of error in the alignments (e.g., false orthologous, segmental duplications, errors in ancestral sequence reconstruction) are still possible and can result in inflated dN/dS. The dN/dS method is not suited for comparing very closely related species and therefore, differences between Neanderthal, Denisovan, and Homo sapiens must be taken with care. Moreover, methods to estimate the evolution of proteins are expected to give downwardly biased estimates (Eyre-Walker and Keightley 2009). However, our GC12 normalization has already proved to correct for most of those biases in systematic analyses (Kapheim et al. 2015), and our raw dN/dS values highly correlate with other independent studies on primates (Nielsen et al. 2005; Biswas et al. 2016). Moreover, for the enrichment analyses, we used bootstrapping techniques to better control for potential biases induced by differences in GC content and gene length, especially for genes implicated in brain disorders (Zylka et al. 2015). Finally, our data are openly available on the companion website and allow checking at the variant level which amino acids changed.
Perspectives
Our systematic analysis of protein sequence diversity confirmed that protein-coding genes relating to brain function are among the most highly conserved in the human genome. The set of PSGs identified here may have played specific roles in the evolution of human cognition, by modulating brain size, neuronal migration, and synaptic physiology, but further genetic—including detailed analyses of all species branches—and functional studies would shed new light on the role of these genes. Beyond the brain, this resource will also be useful for estimating the evolutionary pressure acting on genes related to other biological pathways, particularly those displaying signs of positive selection during primate evolution, such as the reproductive and immune systems.
Methods
Genetic sequences
Alignments with the reference genome
We collected sequences and reconstructed sequence alignments with the reference human genome version hg19 (release 19, GRCh37.p13). For the primate common ancestor sequence, we used the Ensembl 6-way Enredo-Pecan-Ortheus (EPO) (Paten et al. 2008) multiple alignments v71, related to Homo sapiens (hg19), chimpanzee (panTro4), gorilla (gorGor3), orangutan (ponAbe2), rhesus macaque (rheMac3), and marmoset (calJac3). For the two ancestral hominins, Altai, and Denisovan, we integrated variants detected by Castellano et al. (2014) into the standard hg19 sequence (http://cdna.eva.mpg.de/neandertal/; date of access July 3, 2014). Finally, we used the whole-genome alignment of all the primates used in the 6-EPO from the UCSC website (http://hgdownload.soe.ucsc.edu/downloads.html; date of access online August 13, 2015). All the PSGs had their protein sequence deduced from our analysis compared manually to the one in the protein database. All variants matched, and we did not find any alignment artifact. The core annotations used for our study were not available for the GRCh38 version of the human genome when we started this project. Because one of the biggest improvements in GRCh38 is the annotation of the centromere regions (Guo et al. 2017), a switch from GRCh37 to GRCh38 would not affect our conclusions. Moreover, regarding the coding regions of the human genome, the number of nonsynonymous detected by GRCh38 (N = 22,796 SNVs) is very similar to GRCh37's (N = 22,622 SNVs) (Guo et al. 2017, Table 3).
VCF annotation
We combined the VCF file from Castellano et al. (2014) with the VCF files generated from the ancestor and primate sequence alignments. The global VCF was annotated with ANNOVAR (version of June 2015) (Wang et al. 2010), using the following databases: refGene, cytoBand, genomicSuperDups, esp6500siv2_all, 1000g2014oct_all, 1000g2014oct_afr, 1000g2014oct_eas, 1000g2014oct_eur, avsnp142, ljb26_all, gerp++elem, popfreq_max, exac03_all, exac03_afr, exac03_amr, exac03_eas, exac03_fin, exac03_nfe, exac03_oth, exac03_sas. We also used the ClinVar database (https://ncbi.nlm.nih.gov/clinvar/; date of access February 3, 2016).
ωGC12 calculation
Once all the alignments had been collected, we extracted the consensus coding sequences (CCDS) of all protein-coding genes referenced in Ensembl BioMart GRCh37, according to the HGNC (date of access May 5, 2015) and NCBI Consensus CDS protein set (date of access August 10, 2015). We calculated the number of nonsynonymous mutations N, the number of synonymous mutations S, the ratio of the number of nonsynonymous mutations per nonsynonymous site dN, the number of synonymous mutations per synonymous site dS, and their ratio dN/dS (also called ω) between all taxa and the ancestor, using the yn00 algorithm implemented in PamL software (Yang 2007). We avoided infinite and null results by calculating a corrected version of dN/dS. If S was null, we set its value to 1 to avoid having zero as the numerator. The obtained values were validated through the replication of a recent systematic estimation of dN/dS between Homo sapiens and two great apes (Pan troglodytes and Pongo abelii; Pearson's r > 0.8, P < 0.0001) (Supplemental Fig. S2; Biswas et al. 2016). Finally, we obtained our ωGC12 value by correcting for the GC12 content of the genes with a generalized linear model and by calculating a Z-score for each taxon (Kapheim et al. 2015). GC content has been associated with biases in mutation rates, particularly in primates (Galtier et al. 2009) and humans (Kostka et al. 2012). We retained only the 11,667 genes with 1:1 orthologs in primates (extracted for GRCh37.p13 with Ensembl BioMart; accessed online February 27, 2017).
Gene sets
We used different gene sets, starting at the tissue level and then focusing on the brain and key pathways. For body tissues, we used Illumina Body Map 2.0 RNA-seq data, corresponding to 16 human tissue types: adrenal, adipose, brain, breast, colon, heart, kidney, liver, lung, lymph, ovary, prostate, skeletal muscle, testes, thyroid, and white blood cells (for more information: https://www.ebi.ac.uk/gxa/experiments/E-MTAB-513; data preprocessed with Cufflinks, accessed May 5, 2015, at http://cureffi.org). We also used the microarray data set of Su et al. (2004) (Human U133A/GNF1H Gene Atlas, accessed May 4, 2015, at http://biogps.org). Finally, we also replicated our results with recent RNA-seq data from The GTEx Consortium (2015) (https://www.gtexportal.org/home/).
For the brain, we used the data set of Su et al. (2004) and the Human Protein Atlas data (accessed November 7, 2017, at https://www.proteinatlas.org). For analysis of the biological pathways associated with the brain, we used KEGG (accessed February 25, 2015, at https://www.genome.jp/kegg/), synaptic genes curated by the group of Danielle Posthuma at Vrije Universiteit (accessed September 1, 2014, at https://ctg.cncr.nl/software/genesets) (Ruano et al. 2010; Lips et al. 2012), and mass spectrometry data from Loh (2016). Finally, for the diseases associated with the brain, we combined gene sets generated from Human Phenotype Ontology (accessed August 14, 2020, at https://hpo.jax.org) including OMIM annotation (https://omim.org) and curated lists: the 65 risk genes proposed by Sanders et al. (2015) (TADA), the candidate genes for autism spectrum disorders from SFARI (accessed July 17, 2015, at https://gene.sfari.org), the Developmental Brain Disorder (DBD) (accessed July 12, 2016, at https://geisingeradmi.org/care-innovation/studies/dbd-genes/), and Cancer Census (accessed November 24, 2016, at https://cancer.sanger.ac.uk/census) data. Note that the combination of HPO and OMIM is the most exhaustive, making it possible to avoid missing potential candidate genes, but this combination does not identify specific associations.
SynGO was generously provided by Matthijs Verhage (accessed on January 11, 2019). This ontology is a consistent, evidence-based annotation of synaptic gene products developed by the SynGO consortium (2015–2017) in collaboration with the GO consortium. It extends the existing Gene Ontology (GO) of the synapse and follows the same dichotomy between biological processes (BP) and cellular components (CC).
For single-cell transcriptomics data sets, we identified the genes specifically highly expressed in each cell type, following the same strategy as used for the other RNA-seq data sets. The single-cell data for the developing human cortex were kindly provided by Maximilian Haeussler (available at https://cells.ucsc.edu; accessed on October 30, 2018). The single-cell transcriptional atlas data for the developing murine cerebellum (Carter et al. 2018) were kindly provided by Robert A. Carter (accessed on January 29, 2019). For each cell type, we combined expression values cross all available replicates, to guarantee a high signal-to-noise ratio. We then calculated the values for the associated genes in Homo sapiens according to the paralogous correspondence between humans and mice (Ensembl BioMart accessed on February 23, 2019).
Gene nomenclature
We extracted all the Entrez ID of the protein-coding genes for GRCh37 from Ensembl BioMart. We used the HGNC database to recover their symbols. For the 46 unmapped genes, we searched the NCBI database manually for the official symbol.
McDonald–Kreitman test, neutrality index, and Direction of Selection
We assessed the possible fixation of variants in the Homo sapiens population by first calculating the relative ratio of nonsynonymous to synonymous polymorphism (pN/pS) from the 1000 Genomes VCF for all SNPs, for SNPs with a minor allele frequency (MAF) <1% and <5%. SNPs were annotated with ANNOVAR across 1000 Genomes Project (ALL+5 ethnicity groups), ESP6500 (ALL+2 ethnicity groups), ExAC (ALL+7 ethnicity groups), and CG46 (for more details, see http://annovar.openbioinformatics.org/en/latest/user-guide/filter/#popfreqmax-and-popfreqall-annotations). The polymorphism ratio (pN/pS) allowed us to take into account the constraint on nonsynonymous sites and thus increase the power of detecting positive selection (Salvador-Martínez et al. 2018). We indeed normalized the divergence ratio (dN/dS) using the McDonald–Kreitman (MK) test, that is, calculating the neutrality index (NI) as the ratio of raw pN/pS and dN/dS values (McDonald and Kreitman 1991). We considered the PSG to be fixed in the population when NI < 1. We also confirmed with a new statistic for evolutionary measure: the Direction of Selection (DoS) = Dn/(Dn + Ds) − Pn/(Pn + Ps) (Stoletzki and Eyre-Walker 2011) that all divergent genes with NI < 0 had a DoS < 0 (Supplemental Fig. S8).
NeuroVault analyses
We used the NeuroVault website (Gorgolewski et al. 2015) to collect 19,244 brain imaging results from fMRI-BOLD studies (T and Z-score maps) and their correlation with the gene expression data (Gorgolewski et al. 2014) of the Allen Brain Atlas (Hawrylycz et al. 2012). The gene expression data of the Allen Brain Atlas were normalized and projected into the MNI152 stereotactic space used by NeuroVault, using the spatial coordinates provided by the Allen Brain Institute. An inverse relationship between cortical and subcortical expression dominated the pattern of expression for many genes. We thus calculated the correlations for the cortex and subcortical structures separately.
Allen brain data
We downloaded the Allen Brain Atlas microarray-based gene data and multiple cortical areas: Smart-seq from the Allen Brain website (accessed on July, 2020, at http://www.brain-map.org). Microarray data were available for six adult brains; the right hemisphere was missing for three donors, so we considered only the left hemisphere for our analyses. For each donor, we averaged probes targeting the same gene and falling in the same brain area. We then subjected the data to log normalization and calculated Z-scores: across the 20,787 genes for each brain region to obtain expression levels; and across the 212 brain areas for each gene to obtain expression specificity. For genes with more than one probe, we averaged the normalized values over all probes available. The Smart-seq data set followed a similar preprocessing and led to expression level and specificity of 32,165 genes across 363 cell types.
As a complementary data set, we also used a mapping of the Allen Brain Atlas onto the 68 brain regions of the FreeSurfer atlas (French and Paus 2015) (accessed on April 4, 2017, at https://figshare.com/articles/A_FreeSurfer_view_of_the_cortical_transcriptome_generated_from_the_Allen_Human_Brain_Atlas/1439749). The expression and specificity measure were used for the 3D visualization in the companion website.
Statistics
Enrichment analyses
We first calculated a two-way hierarchical clustering on the normalized dN/dS values (ωGC) across the whole genome (Fig. 1B). Of note, 11,667 genes were included in the analysis to ensure medium-quality coverage for Homo sapiens, Neanderthals, Denisovans, and Pan troglodytes (Supplemental Fig. S1). According to 30 clustering indices (Charrad et al. 2014), the best partitioning in terms of evolutionary pressure was into two clusters of genes: SCGs (N = 4825; in HS, mean = −0.88 median = −0.80 SD = 0.69) and PSGs (N = 6842; in HS, mean = 0.60 median = 0.48 SD = 0.63). For each cluster, we calculated the enrichment in biological functions in Cytoscape (Shannon et al. 2003) with the BINGO plugin (Maere et al. 2005). We used all 11,667 genes as the background. We eliminated redundancy by first filtering out all the statistically significant Gene Ontology (GO) terms associated with fewer than 10 or more than 1000 genes, and then combining the remaining genes with the EnrichmentMap plugin (Merico et al. 2010). We used a P-value cutoff of 0.005, an FDR Q-value cutoff of 0.05, and a Jaccard coefficient of 0.5.
For the cell type–specific expression analysis (CSEA) (Xu et al. 2014), we used the CSEA method with the online tool http://genetics.wustl.edu/jdlab/csea-tool-2/. This method associates gene lists with brain expression profiles across cell types, regions, and time periods.
Wilcoxon and rank-biserial correlation
We investigated the extent to which each gene set was significantly more under positive or constraint selection than expected
by chance by performing Wilcoxon tests on the normalized dN/dS values (ωGC) for the genes in the set against zero (the mean value for the genome). We quantified effect size by matched pairs rank-biserial
correlation, as described by Kerby (2014). Following nonparametric Wilcoxon signed-rank tests, the rank-biserial correlation was evaluated as the difference between
the proportions of negative and positive ranks over the total sum of ranks as follows:
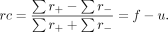 It corresponds to the difference between the proportion of observations consistent with the hypothesis (f) minus the proportion of observations contradicting the hypothesis (u), thus representing an effect size. Like other correlational measures, its value ranges from −1 to +1, with a value of zero
indicating no relationship. In our case, a negative rank-biserial correlation corresponds to a gene set in which more genes
have negative ωGC values than positive values, revealing a degree of conservation greater than the mean for all genes (i.e., ωGC = 0). Conversely, a positive rank-biserial correlation corresponds to a gene set that is more under positive selection than
expected by chance (i.e., taking randomly the same number of genes across the whole genome; correction for the potential biases
for GC content and CDS length are done at the bootstrap level). All statistics relating to Figures 1D and 2, A and B, are summarized in Supplemental Table S3. All those relating to Figure 4 are summarized in Supplemental Table S5.
It corresponds to the difference between the proportion of observations consistent with the hypothesis (f) minus the proportion of observations contradicting the hypothesis (u), thus representing an effect size. Like other correlational measures, its value ranges from −1 to +1, with a value of zero
indicating no relationship. In our case, a negative rank-biserial correlation corresponds to a gene set in which more genes
have negative ωGC values than positive values, revealing a degree of conservation greater than the mean for all genes (i.e., ωGC = 0). Conversely, a positive rank-biserial correlation corresponds to a gene set that is more under positive selection than
expected by chance (i.e., taking randomly the same number of genes across the whole genome; correction for the potential biases
for GC content and CDS length are done at the bootstrap level). All statistics relating to Figures 1D and 2, A and B, are summarized in Supplemental Table S3. All those relating to Figure 4 are summarized in Supplemental Table S5.
Validation by resampling
We also used bootstrapping to correct for potential bias in the length of the coding sequence or the global specificity of
gene expression (Tau, see the methods in Kryuchkova-Mostacci and Robinson-Rechavi 2016). For each of the 10,000 permutations, we randomly selected the same number of genes as for the sample of genes from the
complete set of genes for which dN/dS was not missing. We corrected for CCDS length and GC content by bootstrap resampling. We estimated significance, to determine
whether the null hypothesis could be rejected, by calculating the number of bootstrap draws (Bi) falling below and above the observed measurement (m). The corresponding empirical P-value was calculated as follows:
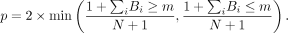
Data access
All the data and code supporting the findings of this study are available from our resource website (https://genevo.pasteur.fr) and as Supplemental Material.
Competing interest statement
The authors declare no competing interests.
Acknowledgments
We thank J.-P. Changeux, L. Quintana-Murci, E. Patin, G. Laval, B. Arcangioli, D. DiGregorio, L. Bally-Cuif, A. Chedotal, C. Berthelot, H. Roest Crollius, and V. Warrier for advice and comments, and we thank the members of the Human Genetics and Cognitive Functions laboratory for helpful discussions. We also thank C. Gorgolewski, R. Carter, M. Haeussler, M. Verhage, and the SynGO consortium for providing key data sets without which this work would not have been possible. This work was supported by the Institut Pasteur; Centre National de la Recherche Scientifique; Paris Diderot University; the Fondation pour la Recherche Médicale (DBI20141231310); the Human Brain Project; the Cognacq-Jay Foundation; the Bettencourt-Schueller Foundation; the Agence Nationale de la Recherche (ANR) (SynPathy); and the Innovative Medicines Initiative 2 [No. 777394]. This research was also supported by the Laboratory of Excellence GENMED (Medical Genomics) Grant No. ANR-10-LABX-0013, Bio-Psy, and by the INCEPTION program ANR-16-CONV-0005, all managed by the ANR part of the Investments for the Future program. G.D. is funded by the Institute for Data Valorization (IVADO), Montreal, and the Fonds de Recherche du Québec (FRQ, 295291; 295289). The funders had no role in study design, data collection, and analysis, the decision to publish, or preparation of the manuscript.
Author contributions: G.D. and T.B. devised the project and came up with the main conceptual ideas. G.D. developed the methods, performed the analyses, and designed the figures. G.D. and T.B. discussed the results and wrote the manuscript. S.M. developed the companion website.
Footnotes
-
[Supplemental material is available for this article.]
-
Article published online before print. Article, supplemental material, and publication date are at https://www.genome.org/cgi/doi/10.1101/gr.262113.120.
-
Freely available online through the Genome Research Open Access option.
- Received February 8, 2020.
- Accepted January 6, 2021.
This article, published in Genome Research, is available under a Creative Commons License (Attribution 4.0 International), as described at http://creativecommons.org/licenses/by/4.0/.