Abstract
Whereas the neurological effects of cocaine have been well documented, effects of acute cocaine consumption on genome-wide gene expression across the brain remain largely unexplored. This question cannot be readily addressed in humans but can be approached using the Drosophila melanogaster model, where gene expression in the entire brain can be surveyed at once. Flies exposed to cocaine show impaired locomotor activity, including climbing behavior and startle response (a measure of sensorimotor integration), and increased incidence of seizures and compulsive grooming. To identify specific cell populations that respond to acute cocaine exposure, we analyzed single-cell transcriptional responses in duplicate samples of flies that consumed fixed amounts of sucrose or sucrose supplemented with cocaine, in both sexes. Unsupervised clustering of the transcriptional profiles of a total of 86,224 cells yielded 36 distinct clusters. Annotation of clusters based on gene markers revealed that all major cell types (neuronal and glial) as well as neurotransmitter types from most brain regions were represented. The brain transcriptional responses to cocaine showed profound sexual dimorphism and were considerably more pronounced in males than females. Differential expression analysis within individual clusters indicated cluster-specific responses to cocaine. Clusters corresponding to Kenyon cells of the mushroom bodies and glia showed especially large transcriptional responses following cocaine exposure. Cluster specific coexpression networks and global interaction networks revealed a diverse array of cellular processes affected by acute cocaine exposure. These results provide an atlas of sexually dimorphic cocaine-modulated gene expression in a model brain.
Cocaine use presents a significant socioeconomic health problem (Kariisa et al. 2019; Substance Abuse and Mental Health Services Administration 2019). Although cocaine use results in arousal and euphoria, side effects include accelerated heart rate, mood swings, difficulty sleeping, loss of appetite, and cognitive distortions. Escalated consumption of cocaine can result in psychosis, cardiovascular disease, and stroke.
The propensity for cocaine use depends on genetic and environmental factors. Whereas much is known about the neurological effects of cocaine, information about genetic variants that are associated with variation in individual susceptibility to psychostimulant use remains incomplete. Furthermore, little is known about acute effects of cocaine consumption on genome-wide gene expression across the brain.
Drosophila melanogaster presents an advantageous model system for systems genetic analyses of cocaine consumption (Kaun et al. 2012). Flies can be reared rapidly in large numbers at low cost in defined genetic backgrounds and under controlled environmental conditions, and about 75% of disease-causing genes in humans have fly orthologs (Pandey and Nichols 2011). The crystal structure of the Drosophila dopamine transporter has been obtained and its binding site can accommodate cocaine (Wang et al. 2015). Exposure of cocaine elicits motor responses that resemble behaviors observed in rodents, and flies develop sensitization to repeated intermittent exposure to cocaine (McClung and Hirsh 1998; Filošević et al. 2018). Dopamine (Bainton et al. 2000), the dopamine transporter (DAT) (Wu and Gu 2003) and the serotonin transporter (SerT) (Corey et al. 1994; Demchyshyn et al. 1994; Borue et al. 2009, 2010) have been implicated in mediating cocaine-induced behaviors in flies (Li et al. 2000; Simon et al. 2009). Consistent with the actions of these neurotransmitters, overexpression of the vesicular monoamine transporter in both dopaminergic and serotonergic neurons decreases the response to cocaine (Chang et al. 2006). Thus, fundamental neural mechanisms affected by exposure to psychostimulants are conserved across phyla, from flies to humans.
Studies on inbred wild-derived, fully sequenced lines of the Drosophila melanogaster Genetic Reference Panel (Mackay et al. 2012; Huang et al. 2014) identified candidate genes associated with variation in consumption and development of preference for cocaine and methamphetamine (Highfill et al. 2019). Targeted RNA interference (RNAi) of gene expression implicated dopaminergic neurons and the mushroom bodies, central brain structures associated with experience-dependent modification of behavior, with consumption and development of preference for these psychostimulants (Highfill et al. 2019). However, RNAi-mediated reduction in expression of candidate genes in glia also affected cocaine consumption, suggesting that widespread brain regions contribute to cocaine-associated behavioral phenotypes. The present study aims to delineate the effects of acute cocaine consumption on genome-wide gene expression across the Drosophila brain.
Results
Cocaine consumption causes behavioral impairments
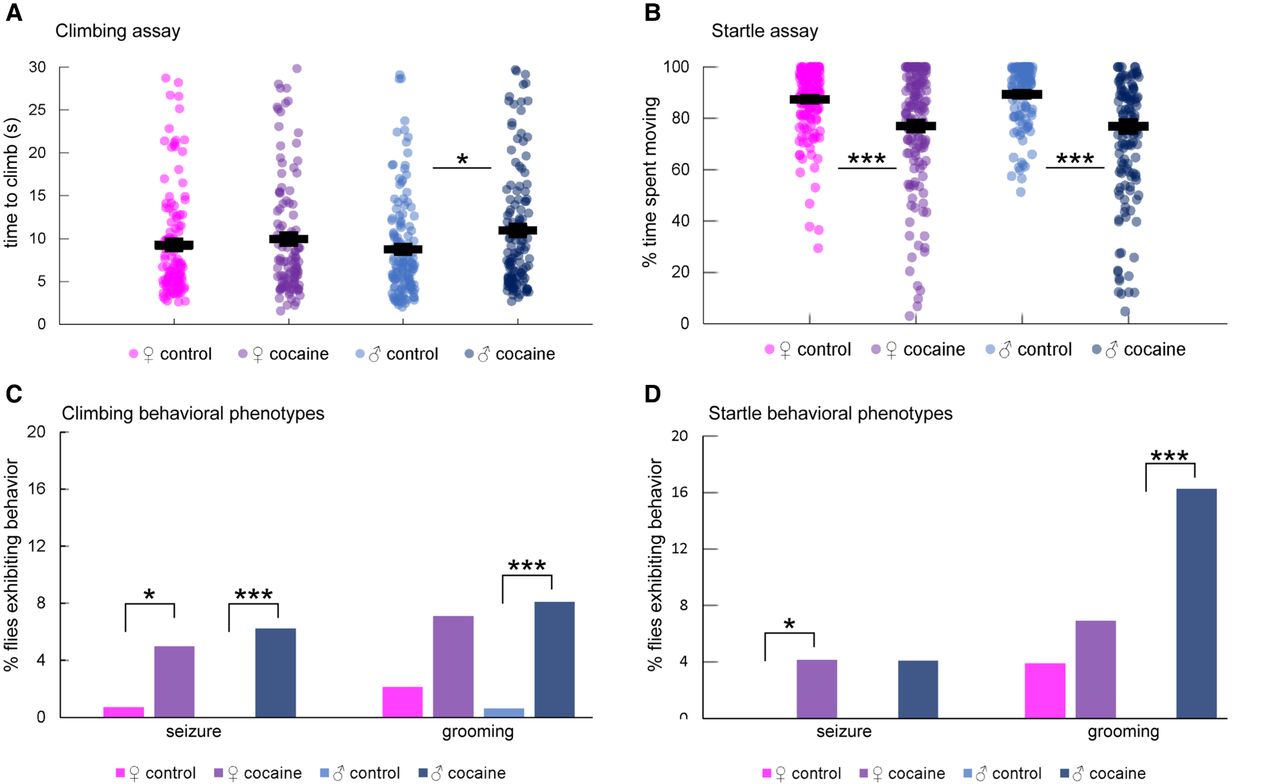
To assess the effects of acute cocaine exposure on fly behavior, we allowed males and females to ingest a fixed amount of sucrose or sucrose supplemented with cocaine within a maximal 2-h time period. We measured negative geotaxis, an innate locomotor behavior, to assess locomotion impairments, and startle behavior as a measure of sensorimotor integration (Fig. 1; Supplemental Table S1). Male flies exposed to cocaine took longer to climb in the negative geotaxis assay than control flies, whereas females appeared unaffected (Fig. 1A). Both male and female flies exposed to cocaine spent less time moving after being subjected to a mechanical disturbance (Fig. 1B; Supplemental Video S1). The average reduced locomotor activity in both assays might result from excessive grooming behavior in a fraction of male flies exposed to cocaine (Fig. 1C,D; Supplemental Video S2). In addition, we observed seizures in a small percentage of flies after cocaine intake during the negative geotaxis assay (Supplemental Video S3). Seizures rarely occurred in controls (Fig. 1C,D). Collectively, these experiments provide evidence that acute exposure to cocaine results in neurological impairments.
Behavioral characterization of Canton S (B) flies after cocaine exposure. (A) Negative geotaxis. The 7.5-cm climb time for each fly was measured. n = 120 (♀control), 114 (♀cocaine), 141 (♂control), 128 (♂cocaine). Horizontal lines represent means with standard error. Male flies exposed to cocaine took longer on average to climb compared to controls. (*) P = 0.0042; two-tailed Student's t-test. (B) Startle response. The percent time out of 45 sec that each fly spent moving following a 42-cm drop was measured. n = 155 (♀control), 145 (♀cocaine), 120 (♂control), 123 (♂cocaine). Horizontal lines represent means with standard error. Flies exposed to cocaine spent less time moving on average than controls. For females, (***) P = 4.68 × 10−6; for males, (***) P = 7.62 × 10−13; two-tailed Student's t-test. (C) Seizures and grooming activity during negative geotaxis. The percent of flies that exhibited seizures or grooming activity during the negative geotaxis assay after exposure to cocaine was measured. n = 142 (♀control), 141 (♀cocaine), 166 (♂control), 161 (♂cocaine). Both females and males exposed to cocaine exhibited seizure activity more than controls. For females, (*) P = 0.0361; for males seizure, (***) P = 0.0007; Fisher's exact test. Males exposed to cocaine also exhibited excessive grooming activity more than controls (grooming: [***] P = 0.0007; Fisher's exact test), but females did not show statistically significant differences. (D) Seizures and grooming activity during the startle response. The percent of flies that exhibited seizures or grooming activity during the startle assay after exposure to cocaine was measured. n = 155 (♀control), 145 (♀cocaine), 120 (♂control), 123 (♂cocaine). Female flies exposed to cocaine exhibited more seizure activity than controls ([*] P = 0.0121; Fisher's exact test), whereas male flies exposed to cocaine exhibited more grooming activity than control ([***] P = 0.00001; Fisher's exact test).
Single-cell RNA-seq reveals cocaine-modulated gene expression in neurons and glia
To assess effects of cocaine consumption on brain gene expression, we analyzed single-cell transcriptional responses in duplicate samples of flies that consumed fixed amounts of sucrose or sucrose supplemented with cocaine in both males and females (Supplemental Table S2). Visualization of the resulting integrated data set using the Uniform Manifold Approximation and Projection (UMAP) nonlinear dimensionality reduction method (Becht et al. 2019) showed that no single cluster was dominated by a specific sample, sex, condition, or replicate and that there was considerable homogeneity (i.e., an even distribution of cells from samples) across the entire data set (Supplemental Fig. S1). We identified 691 differentially expressed genes in males and 322 in females following acute exposure to cocaine, of which ∼69% have human orthologs (Supplemental Table S3). Unsupervised clustering of the integrated data set based on the expression profiles of individual cells resulted in 36 distinct, stable clusters (Fig. 2). We assessed the stability of clustering by examining the relationship between the number of new clusters identified and the granularity resolution parameter (Butler et al. 2018). At a resolution of 0.8, the number of clusters stabilized and the resolution had to be increased significantly from this value in order to add new clusters, indicating that saturation in the diversity of expression profiles had been reached.
UMAP visualization and clustering of single-cell expression data. Cells were clustered based on their expression pattern using the unsupervised shared nearest neighbor (SNN) clustering algorithm. Individual dots represent each cell and the colors of the dots represent the cluster to which the cells belong. Identification of cell types from clusters was performed by cross-referencing cluster-defining genes across FlyBase (Thurmond et al. 2019) and published literature (see Supplemental Table S4).
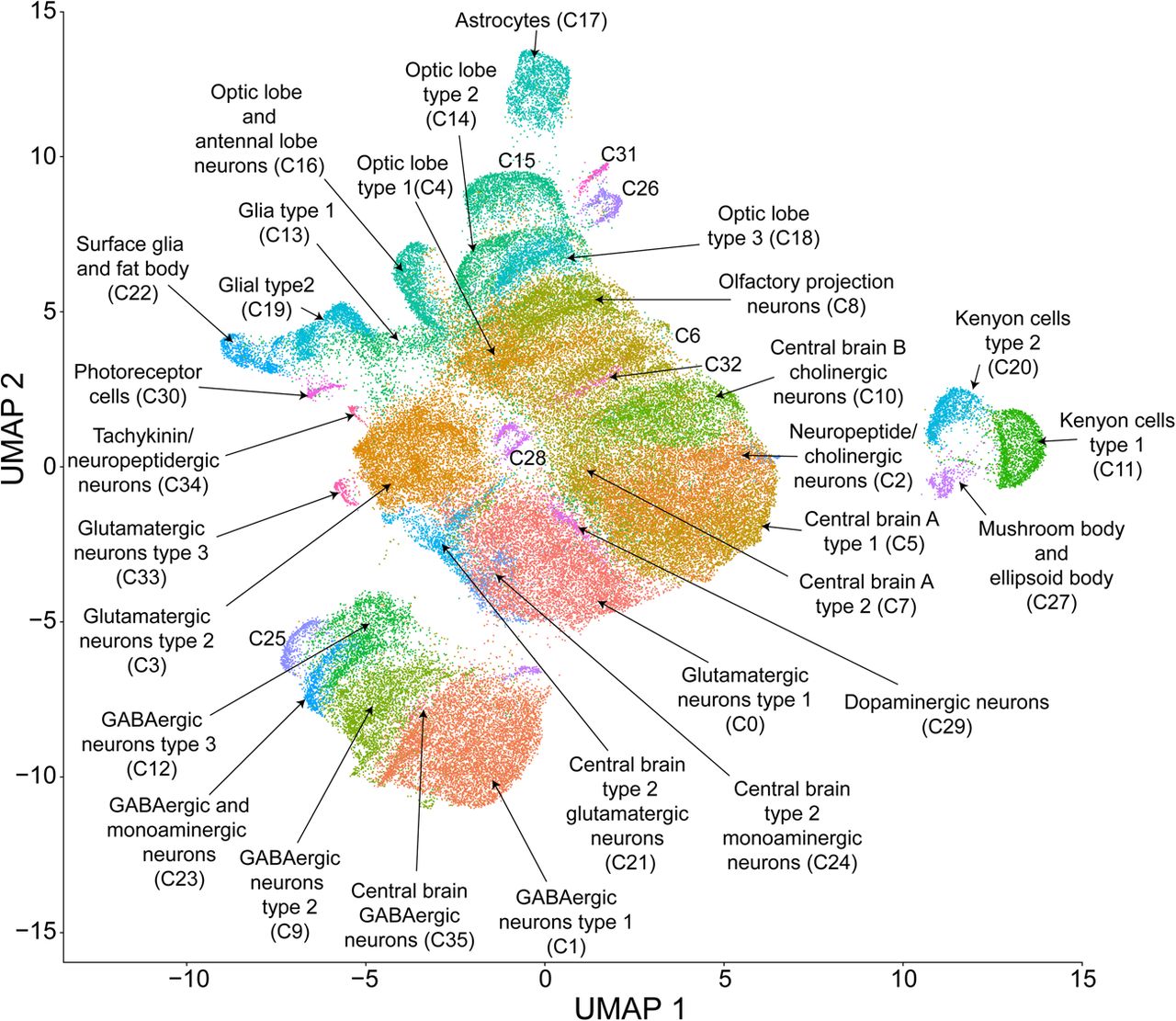
We identified cell types corresponding to each cluster using the top marker genes from each cluster, obtained by comparing each cluster's gene expression profile against the rest of the data set and filtered by |logeFC| > 0.5, Bonferroni-adjusted P-value < 0.05. Annotation of clusters based on their gene markers revealed that all major cell types (neuronal and glial) as well as neurotransmitter types from most brain regions, including the mushroom bodies, were represented (Fig. 2; Supplemental Table S4). Differential expression analysis within individual clusters indicated cluster-specific transcriptional responses to cocaine. Especially, clusters corresponding to glia and Kenyon cells of the mushroom bodies showed transcriptional responses following cocaine exposure (Fig. 3A,B). Thus, acute exposure to cocaine elicits rapid widespread changes in gene expression throughout the brain.
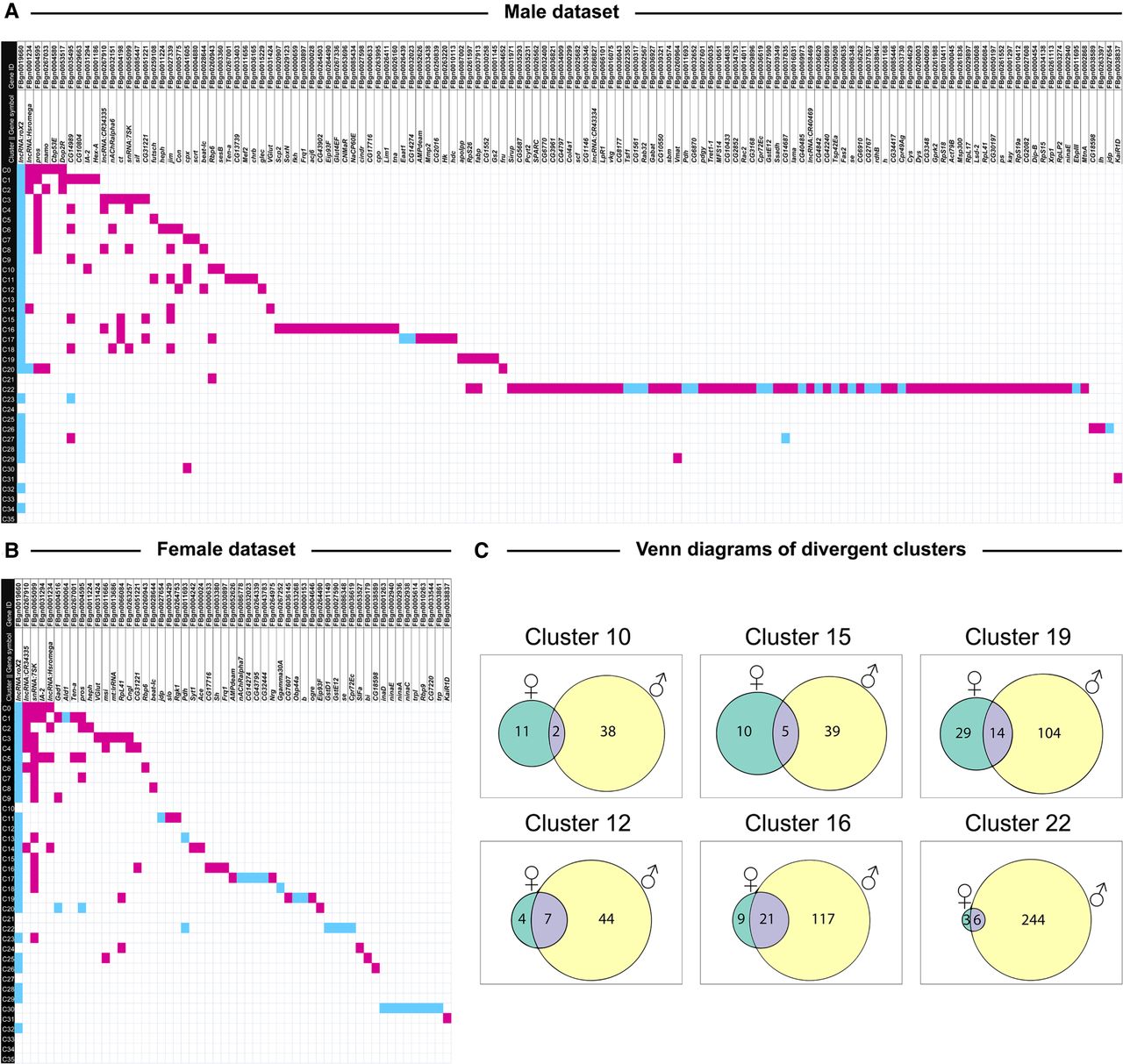
Distribution of differentially expressed genes across clusters in males (A) and females (B) exposed to cocaine, and Venn diagrams showing overlap between differentially expressed genes in males and females (C). To identify clusters with unique gene expression patterns following acute exposure to cocaine, we filtered the list of differentially expressed genes to only show the strongest responses (|logeFC| > 1.0, Bonferroni-adjusted P-value < 0.05) to construct an expression matrix. Differentially expressed genes are listed on the top (columns) and cell clusters are represented by the rows. Magenta boxes show up-regulation and turquoise boxes show down-regulation of gene expression as a result of exposure to cocaine. Panel C shows Venn diagrams of clusters with sexually dimorphic responses to cocaine exposure. The numbers within each Venn diagram represent the unique and shared differentially expressed (|logeFC| > 0.5, Bonferroni-adjusted P-value < 0.05) genes due to cocaine exposure from DGE analysis performed for the corresponding cluster in male and female data sets separately.
Cocaine-modulated changes in gene expression are sexually dimorphic
We first analyzed differential expression by combining the male and female samples. There was a significantly greater number of genes up-regulated than down-regulated across all clusters in response to cocaine. Based on the number of strongly differentially expressed genes (|logeFC| > 1, Bonferroni-adjusted P-value < 0.05), clusters corresponding to surface glia (C22), unannotated cluster C16, astrocytes (C17), and Kenyon cells (C11) showed the largest responses to cocaine (Supplemental Fig. S2). In addition, a core set of genes, selected based on their ranks from the TopKLists consistency analysis (Schimek et al. 2015), show that they responded globally to cocaine exposure (Supplemental Table S5). These genes include: Rpl41, IA-2, and the long noncoding RNAs CR34335 and mt:lrRNA, which were up-regulated; and roX2 and ninaE, which were down-regulated after consumption of cocaine (Supplemental Fig. S2; Supplemental Tables S5, S6).
Examination of males and females separately revealed extensive sexual dimorphism in the response to cocaine. Consistent with effects on organismal phenotypes (Fig. 1), males showed more widespread changes in cocaine-modulated transcript abundances than females (Fig. 3A,B), When we consider only highly differentially expressed genes (|logeFC| > 1.0, Bonferroni-adjusted P-value < 0.05), we can construct expression matrices of 133 genes in males (Fig. 3A) and 54 genes in females (Fig. 3B). Clusters C11 (Kenyon cells), C16 (not annotated), C17 (astrocytes), and C22 (surface glia) had unique responses compared to the rest of the clusters in both males and females; C22 shows the most extensive cocaine-induced changes in transcript abundances in males (Fig. 3A). In addition to differences in the magnitude of cocaine-modulated gene expression between the sexes, we also observe differences in direction, in which up-regulation in one sex corresponds to down-regulation in the other. Overall, there was little overlap between the sexes—especially in clusters C10, C12, C15, C16, C19, and C22 (Fig. 3C; Supplemental Tables S7–S9). Thus, although cocaine-modulated changes in gene expression are widespread throughout the brain in both sexes, specific changes in transcript abundances are distinct between males and females.
Coexpression networks highlight effects of cocaine on diverse cellular processes
Reactome analysis of pathway enrichment (Fabregat et al. 2018) in C11 and C20 in females, which, based on biomarkers, represent Kenyon cells of the mushroom bodies, highlighted inositol phosphate metabolism (Supplemental Table S10), suggesting a role for cocaine in modulating signal transduction. We were unable to assign a specific identity to C16, which might be comprised of a mixture of neurons from the antennal lobe and optic lobe (Supplemental Table S4). However, pathway analysis of male C16 revealed enrichment of multiple signal transduction pathways, including pathways related to G protein-coupled receptor signaling, activation of serotonin and AMPA- and NMDA-type glutamate receptors, activation of axonal growth inhibition, and Class A/1 Rhodopsin-like receptor signaling (Supplemental Table S10).
In contrast to the signal transduction elements associated with the neuronal C11 and C16 clusters, different cellular mechanisms are associated with cocaine exposure in C17 and C22, which represent astrocytes and surface glia, which comprise the blood-brain barrier in the fly, respectively (Supplemental Table S10). Functional enrichment analysis yielded few differentially expressed genes for females, but for C22 in the male data set, it revealed enrichment of Notch activation and signaling, degradation of GABA, immune pathways related to NF-kB activation, cytokine production and Toll-like receptor signaling, and nonsense-mediated decay and translation initiation (Supplemental Table S10). These observations are in line with expected functions of glia (Kremer et al. 2017).
We used Random Matrix Theory (RMT) (Gibson et al. 2013) to construct coexpression networks of cocaine-modulated differentially coexpressed genes for selected clusters with enough differentially expressed input genes. Across all cell clusters, we find genes of unknown function and genes encoding long noncoding RNAs, which are likely to play a regulatory role (Everett et al. 2020). We present examples of coexpression networks for C16 males (Fig. 4A–C; Supplemental Fig. S3) and C22 males (Fig. 4D; Supplemental Fig. S4).
Subnetworks from coexpression network analyses of DEGs from the male C16 and C22 clusters. Networks are constructed from Pearson coefficient-based coexpression values calculated from scaled data of genes that were differentially expressed (filtered for |logeFC| > 0.5, Bonferroni-adjusted P-value < 0.05) due to cocaine exposure. Coexpressions have been filtered using Random Matrix Theory. (A–C) MCODE subnetworks derived from the full network of male cluster C16. The inset in A corresponds to a subset of genes within the subnetwork that have very strong correlation coefficient values with each other compared to the rest of the data set. Colors of the dots represent the connectivity index derived from MCODE scores. Colors of edges represent the positive (red) and negative (green) correlations. (D) Coexpression network analysis of DEGs from the male C22 cluster.
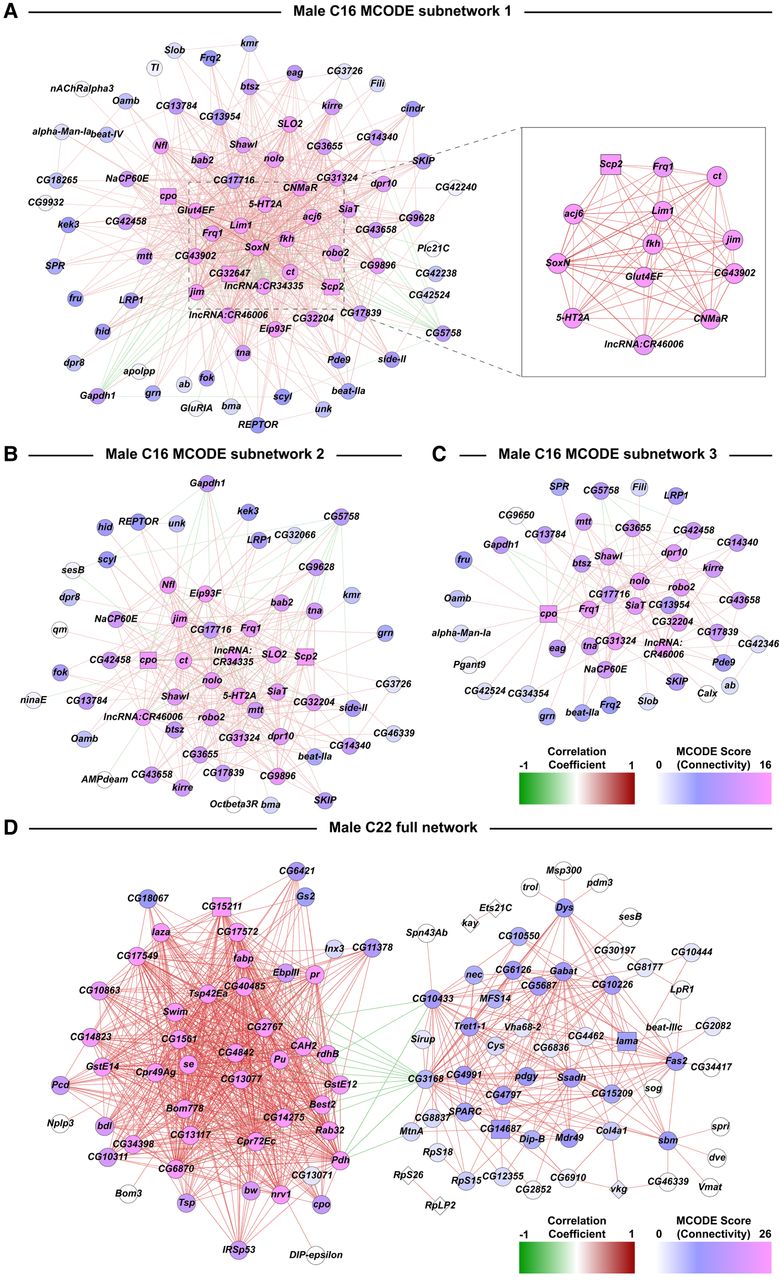
Coexpression analysis for C16 reveals a highly interconnected network that could be partitioned into three subnetworks using Molecular Complex Detection (MCODE) stratification (Bader and Hogue 2003). Central genes include transcriptional regulators associated with development, including dendrite morphogenesis (Lim1, jim) as well as signal transduction (5-HT2A, CNMaR) (Fig. 4A; Supplemental Fig. S3). Random Matrix Theory identified two major subnetworks within the interaction network that represents C22 (Fig. 4D; Supplemental Fig. S4). The two subnetworks in C22 were connected by only three genes (CG3168, CG10433, and CG15209) through negative correlation (Fig. 4D; Supplemental Fig. S4). Of the three genes, CG3168, which belongs to the SLC22 family of organic ion transporters, had the largest number of negative correlations linking the two large subnetworks. This gene is expressed in the blood-brain barrier of flies and postulated to play a role in chemoprotection of the brain (Hindle and Bainton 2014). Further stratification using the MCODE algorithm resulted in three tightly clustered C22 subnetworks. The C22 male interaction network comprises genes associated with oxidation-reduction (se, Ssadh) and redox reactions, particularly the glutathione system (GstE12, GstE14, se), as well as cell adhesion (SPARC, bdl, Tsp, Fas2).
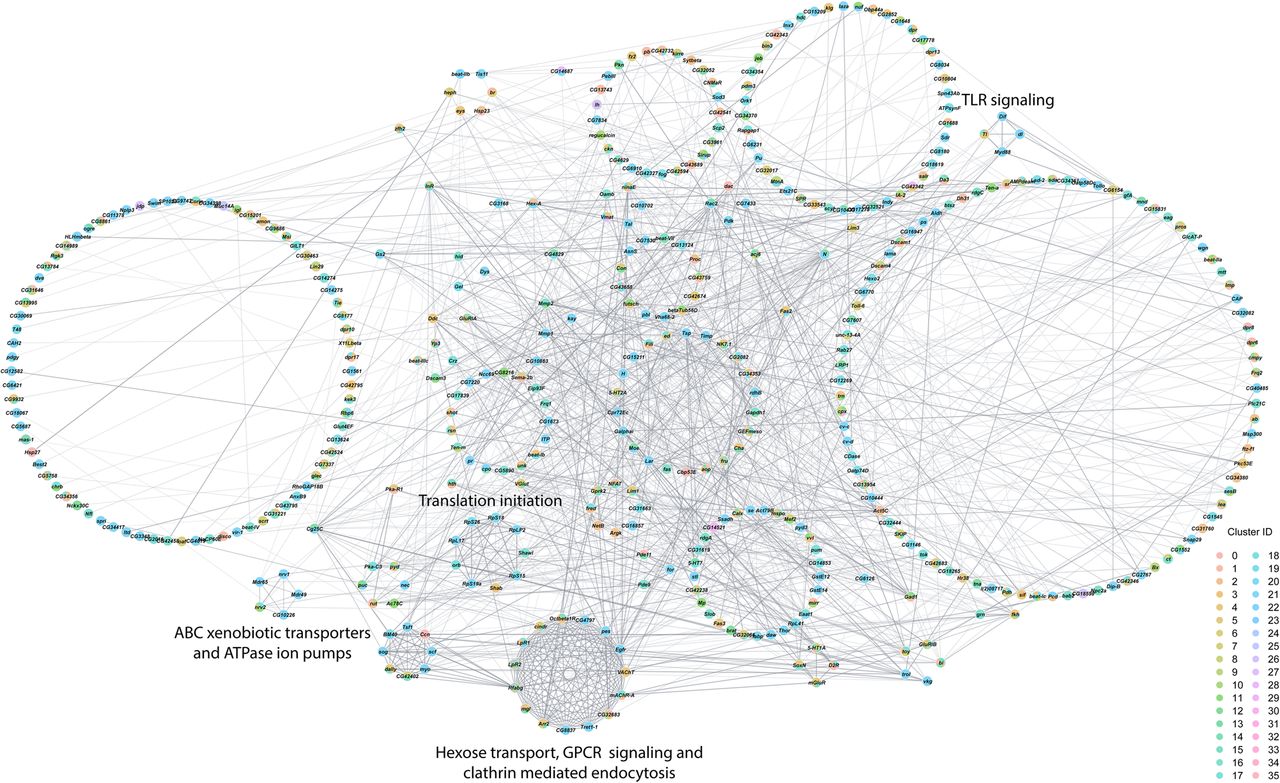
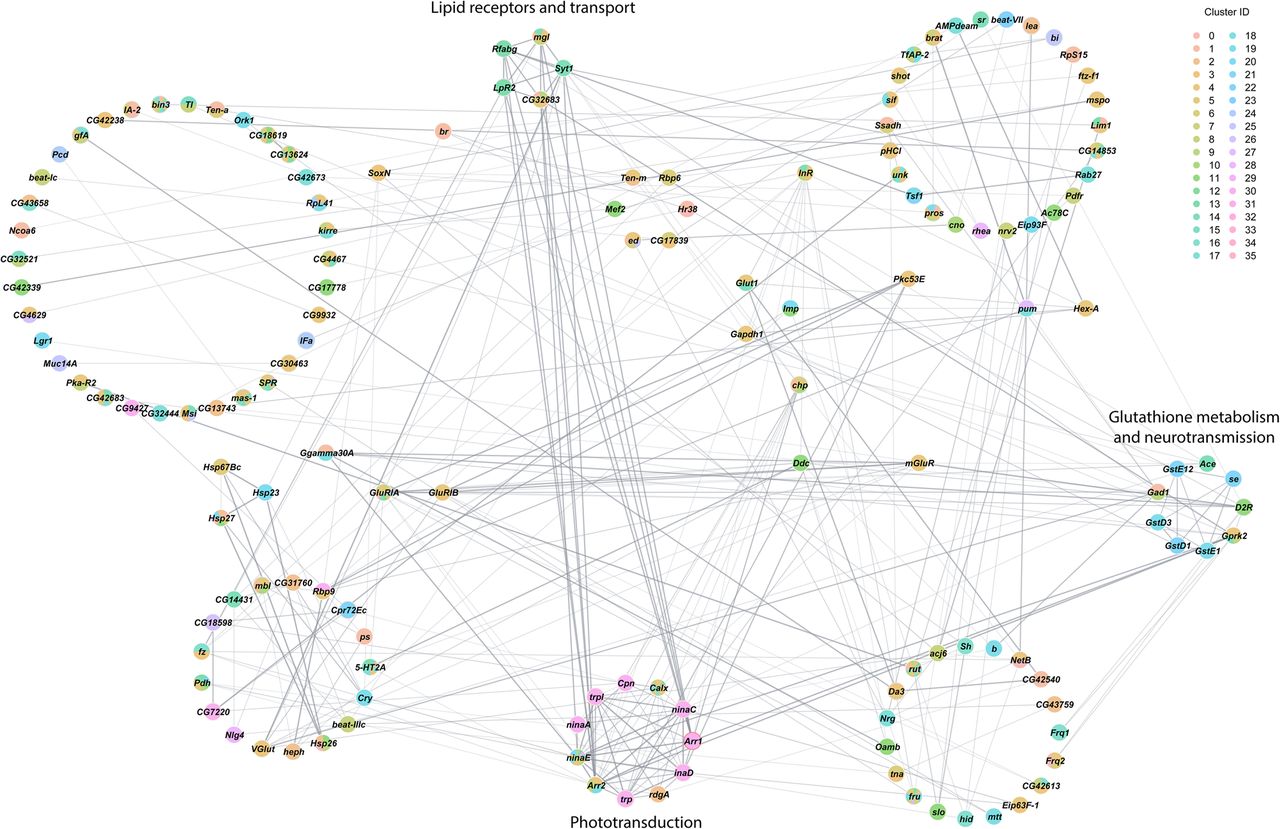
Finally, we assessed interaction networks among differentially expressed genes across all cell clusters separately for males and females (Figs. 5, 6). The global transcriptional response to cocaine in males is captured by a complex network of interconnected modules (Fig. 5). Functional analyses reveal modules associated with Toll-like receptor signaling, ABC xenobiotic transporters and ATPase ion pumps, translation initiation, and hexose transport, G protein-coupled receptor signaling and clathrin-mediated endocytosis. The female network has fewer genes and contains modules associated with phototransduction, lipid receptors and transport, and glutathione metabolism and neurotransmission (Fig. 6). In each network, multiple cell clusters contribute to the organization of each network module, indicating that the transcriptional response to cocaine is coordinated not only within but also across different cells throughout the brain.
Interaction network analysis of DEGs from all clusters in the male data set. Network constructed from interactions calculated using StringApp plugin within Cytoscape for genes that were differentially expressed (filtered for |logeFC| > 0.1 and Bonferroni-adjusted P-value < 0.05) in all clusters from the male data set. Gray edges represent interactions. Genes that were differentially expressed in multiple clusters are depicted as pie charts with each color representing the respective cluster. Genes are grouped into circles based on their MCODE connectivity scores. Annotations of these circular groups represent the pathways that are enriched for the genes within these groups. Bonferroni-adjusted P-value < 0.05 was considered as significant for enrichment in the statistical overrepresentation tests.
Interaction network analysis of DEGs from all clusters in the female data set. Network constructed from interactions calculated using StringApp plugin within Cytoscape for genes that were differentially expressed (filtered for |logeFC| > 0.1 and Bonferroni-adjusted P-value < 0.05) in all clusters from the female data set. Gray edges represent interactions. Genes that were differentially expressed in multiple clusters are depicted as pie charts with each color representing the respective cluster. Genes are grouped into circles based on their MCODE connectivity scores. Annotations of these circular groups represent the pathways that are enriched for the genes within these groups. BH-FDR adjusted P-value < 0.05 was considered as significant for enrichment in the statistical overrepresentation tests.
Discussion
Unlike humans and rodent models of substance abuse, Drosophila enables comprehensive single-cell transcriptomics analyses of living cells across the entire brain in a single analysis (Davie et al. 2018). We generated an atlas of cocaine-modulated gene expression changes in the fly brain and found that transcriptional changes in response to acute consumption of cocaine are rapid, widespread in both neurons and glia, and sexually dimorphic. We performed the experiments in duplicate to establish cross-validation. Transcript abundance levels are influenced by circadian time (Claridge-Chang et al. 2001; Sivachenko et al. 2013; Krzeptowski et al. 2018). Therefore, we performed all experiments within a defined window of circadian time. Because the transcriptional profiles we obtained provide a single ‘snapshot’ in time, we cannot extract information about the temporal progression of the transcriptional response to cocaine to determine whether or to what extent transcriptional responses in different cell populations occur sequentially or in parallel. Also, we cannot draw inferences as to which changes in gene expression might lay a foundation for development of cocaine preference (Highfill et al. 2019), because we only assessed acute transcriptional responses following a single exposure to cocaine.
The response to cocaine is sexually dimorphic
Previous studies on the Drosophila melanogaster Genetic Reference Panel (Mackay et al. 2012; Huang et al. 2014) have documented sexual dimorphism in the genetic architectures of a wide range of morphological (Zwarts et al. 2015), life history (Nuzhdin et al. 1997; Huang et al. 2020), and behavioral traits (Shorter et al. 2015; Harbison et al. 2019), including cocaine consumption and preference (Highfill et al. 2019). However, sexual dimorphism in the transcriptional response following acute exposure to cocaine is extreme compared to any previous studies and is mirrored and consistent with the behavioral phenotypes (Fig. 1). It is possible that differences in cocaine metabolism between males and females may contribute to this sexual dimorphism. The sexual dimorphism we observe is in line with previous studies that show reduced locomotion and increased grooming in flies given low doses of cocaine, with males showing greater impairments in behavior (McClung and Hirsch 1998). We note, however, that changes in gene expression are not, a priori, necessary for cocaine to elicit behavioral effects.
Cocaine-modulated gene expression in the mushroom bodies
Transcriptional effects of cocaine exposure are evident in all cell clusters, but among neuronal populations, the Kenyon cells of the mushroom bodies (represented by C11 and C20) have especially large responses to cocaine. The mushroom bodies are integrative centers associated with experience-dependent modulation of behavior and have previously been implicated in development of preference for cocaine intake (Highfill et al. 2019). Acute cocaine consumption leads to changes in gene products associated with signal transduction, including phosphatidyl inositol-mediated signaling in C11 in males (Supplemental Table S10), as well as cyclic AMP-mediated signaling, which is evident from increased expression of rutabaga (rut), in C20 in females (Supplemental Tables S8, S10). rut encodes a calcium-calmodulin-dependent adenylyl cyclase, implicated in learning and memory (Levin et al. 1992) and behavioral responses to ethanol (Rodan et al. 2002; Xu et al. 2012). cpx, which is involved in the functioning of the SNARE complex at the synapse (Scholz et al. 2019) and may affect neurotransmission, is up-regulated in males in response to cocaine. Kenyon cells receive dopaminergic input, and acute exposure to cocaine results in altered expression of Ddc, which encodes Dopa decarboxylase, and down-regulation of Dop2R, which encodes a dopamine receptor in C11 in females (Supplemental Table S8). slo, which regulates neurotransmitter release at synapses (Jepson et al. 2014), is up-regulated in response to cocaine, as is Rgk1, which plays a role in negative regulation of calcium channel activity (Murakami et al. 2017). Down-regulation of jdp can lead to dopamine blockade through its activity as a cochaperone in synaptic vesicle release (Ye et al. 2004). Based on the collective data, cocaine-induced modulation of gene expression appears to result in altered synaptic regulation in the mushroom bodies.
Cocaine-modulated gene expression in glia
In addition to cocaine-modulated changes in gene expression in neurons, acute exposure to cocaine results in altered transcript abundances in different populations of glia (C13, C17, C19, C22), including surface glia (C22) and astrocytes (C17). Mutants of moody, which encodes two G protein-coupled receptor isoforms localized to surface glia, have increased sensitivity to cocaine (Bainton et al. 2005). The surface glia, represented by perineurial and subperineurial glia, act as the blood-brain barrier (DeSalvo et al. 2014; Kremer et al. 2017) and mediate the innate immune response (Kounatidis and Chtarbanova 2018). Genes associated with the blood-brain barrier in Drosophila are also up-regulated in response to cocaine. This includes ogre, which regulates gap junction channel activity (Holcroft et al. 2013; Spéder and Brand 2014), and Nrg, which plays a role in cell adhesion in the blood-brain barrier (Kanda et al. 2019), in females.
Acute exposure to cocaine causes changes in expression of genes involved with Toll-like receptor (TLR) signaling, nuclear factor kappa B (NF-kB) activation, cytokine production, and glutathione metabolism (Figs. 5, 6). TLR signaling has been associated with response to cocaine (Zhu et al. 2018), likely due to the interaction of cocaine with TLR on microglia (Northcutt et al. 2015), and cocaine activates the NF-kB pathway in the nucleus accumbens of mice (Russo et al. 2009; Muriach et al. 2010).
Astrocytes provide metabolic support for neurons (Tsacopoulos and Magistretti 1996) and regulate neuronal NMDA receptors and synaptic plasticity (Haydon et al. 2009). Glutamatergic neurons feature prominently in C1, C3, C21, and C33. Studies on rats have shown that cocaine is toxic to astrocytes and that loss of astrocytes leads to dysfunctional neuron-glia communication (Badisa et al. 2013, 2014, 2015). Eaat1, which is highly expressed in astrocytes (Supplemental Table S4), encodes a transmembrane glutamate transporter involved in glia-neuron communication (MacNamee et al. 2016). Eaat1 is down-regulated in response to cocaine in males and has been associated with lifespan (Mazaud et al. 2019), long-term memory (Matsuno et al. 2019), seizures (Jen et al. 2005; Cho et al. 2018), and ataxia (Jen et al. 2005; Parinejad et al. 2016). Episodic ataxia due to a mutation in this gene (Parinejad et al. 2016) suggests that altered expression of Eaat1 in astrocytes could play a role in cocaine-induced locomotor effects.
Translating findings from the Drosophila model to cocaine sensitivity in humans
Like flies, people show sexually dimorphic effects of cocaine use. Although substance use disorders are more prevalent in males, females are more likely to escalate their drug usage to the compulsive stage faster than males (Haas and Peters 2000; Westermeyer and Boedicker 2000), report more negative effects during withdrawal, and have greater relapse than males (Becker and Koob 2016). Females metabolize cocaine faster than males, as is evident from lower levels of cocaine metabolites in the bloodstream of females compared to males who have consumed equal amounts of cocaine (Lukas et al. 1996). In this same study, males experienced the effects of cocaine faster and with greater intensity than females. In rats, differences in sex hormones and the estrous cycle contribute to differences in sensitivity to cocaine (Becker and Koob 2016; Cao et al. 2018). Thus, sexual dimorphism is a universal feature of the physiological response to cocaine.
Although the Drosophila brain is anatomically distinct from the vertebrate brain, fundamental neural processes are evolutionarily conserved. Functions of the dopamine reward pathway in humans are analogous to experience-dependent modulation of behaviors by the mushroom bodies. In our study, ∼69% of genes differentially expressed in response to cocaine have human orthologs (Supplemental Table S3), including Aldh, Dop2R, GluRIA, GluRIB, and Vmat, previously implicated in cocaine phenotypes. Polymorphisms in the human ortholog for Aldh, ALDH2, have been associated with drug addiction in Chinese populations (Zhang et al. 2017), and suppression of ALDH2 suppresses cocaine-seeking behavior (Yao et al. 2010). Dop2R encodes a dopamine receptor, and its human ortholog, DRD2, is a well-characterized component of the dopamine reward pathway, which mediates development of cocaine dependence (Noble et al. 1993; Persico et al. 1996; Moyer et al. 2011; Stolf et al. 2019). The glutamate receptor genes GluRIA and GluRIB are associated with glutamatergic neurotransmission, which is altered following exposure to cocaine and has been linked to cocaine sensitization and cocaine-induced behavioral effects (Ghasemzadeh et al. 2011; Liu et al. 2014; Shin et al. 2016). Vmat encodes the vesicular monoamine transporter responsible for packaging the neurotransmitters dopamine, serotonin, and octopamine in synaptic vesicles (Greer et al. 2005). In humans, the solute carrier family 18 member A2, SLC18A2 (previously known as VMAT2), has the same function, and protein levels are reduced in cocaine users (Little et al. 2003; Narendran et al. 2014). These functional parallels between the fly model and human studies provide proof-of-principle that results from cocaine exposure obtained from the fly model can be translated to human populations. Thus, the comprehensive documentation of cocaine-mediated modulation of gene expression which we have derived can serve as a contextual framework for future human studies.
Methods
Drosophila stock
Canton S (B) flies (Norga et al. 2003) were maintained on standard cornmeal/yeast/molasses-agar culture medium at 25°C on a 12:12 h light:dark cycle with 50% humidity in controlled adult density vials to prevent overcrowding. Briefly, five males and five females were placed into a vial and allowed to mate for 2 d before being cleared. Progeny from these vials were collected after eclosion and aged for 3–5 d before experimentation.
Cocaine exposure
Cocaine.HCl was obtained from the National Institute on Drug Abuse under Drug Enforcement Administration license RA0443159. To expose flies to cocaine, we performed a modified version of the capillary feeder (CAFE) assay (Ja et al. 2007). We collected the first 40 flies that consumed 0.53 µL of cocaine and the first 40 flies that consumed 0.53 µL of sucrose, corresponding to an 8-mm reduction in the height of the solution in the capillary. All experiments were carried out between 8 a.m. and 11 a.m. Flies were allowed to feed for no more than 2 h.
Behaviors
We measured negative geotaxis and startle response of individual flies within a 10-min time frame immediately following acute exposure to cocaine in the CAFE assay. We quantified grooming and seizures in addition to measuring the behavioral response in each assay. Excessive grooming was defined as more than 10 sec of constant grooming (Supplemental Video S2). Seizure activity was defined as severe muscle tremors that prevented the fly from moving normally (Supplemental Video S3).
Brain dissection and dissociation
Brains were dissected from each fly immediately after it consumed the designated amount of sucrose or cocaine solution, and we used a dissociation protocol modified from Croset et al. (2018) and Davie et al. (2018). We collected eight samples of 20 brains from males and females exposed to cocaine or sucrose, with two biological replicates per treatment and sex. We proceeded with GEM generation using the Chromium controller (10x Genomics) if we had a live cell count of >500 live cells/µL.
Library preparation and sequencing
We made libraries after GEM generation in accordance with 10x Genomics v3.1 protocols. We sequenced the final libraries on an S1 flow cell using a NovaSeq (Illumina, Inc.) according to the manufacturer's instructions.
FASTQ generation, demultiplexing, and alignment
The mkfastq pipeline within Cell Ranger v3.1 (10x Genomics) was used to convert BCL files from the sequence run folder to demultiplexed FASTQ files. Release 6 version of the Drosophila melanogaster reference GCA_000001215.4 from the NCBI GenBank database (https://www.ncbi.nlm.nih.gov/genbank/) was indexed using the mkref pipeline and used for alignment using the count pipeline within Cell Ranger v3.1 with the expected cell count parameter set to 5000 cells. The sequencing and alignment summary is given in Supplemental Table S2.
Preprocessing, integration, and cell type clustering
Raw expression counts output for each sample from the Cell Ranger pipeline was imported and analyzed using the Seurat v3 package in R (R core team 2013; Butler et al. 2018). Genes expressed in less than five cells and cells with less than 300 or greater than 2500 RNA features were filtered out. The upper (2500) and lower (300) thresholds for the RNA features per cell were chosen based on the recommendations from the developers of the Seurat v3 pipeline. The recommendation is based on the multitude of observations indicating that cells with less than 300 RNA features tend to have very sparse and unreliable signal and those with greater than 2500 RNA features tend to be miscalled multiplet cells. Normalization and subsequent integration were performed using the sctransform pipeline (Hafemeister and Satija 2019). To identify the cell type clusters within the data set, unsupervised clustering using the FindClusters function and a resolution of 0.8 were used. Cluster marker genes were identified using the FindAllMarker function (min.pct equals; 0.25, logfc.threshold equals; 0.5, only.pos equals; TRUE). The top three genes with positive expression for each cluster were extracted and used for cell type characterization.
Differential expression
Differential expression was performed for each cluster in two ways: (1) after combining male and female samples together to test for effects of cocaine that are common to both sexes; and (2) testing for effects of cocaine in males and females separately to identify sexually dimorphic responses. The Pearson residuals output from the sctransform pipeline was used as input for differential expression (DE) calculation (Hafemeister and Satija 2019). The MAST algorithm was used as the testing methodology in the FindMarkers function (test.use equals; “MAST”, assay equals; “SCT”, slot equals; “scale.data”) for each cluster to calculate DE. Clusters with a sufficient number of DEGs were subjected to pathway enrichment analysis using the statistical overrepresentation test using PANTHER (Thomas et al. 2003) and Reactome databases (Fabregat et al. 2016). Pathways with BH-FDR adjusted P-values < 0.05 were considered statistically enriched.
Simulation of bulk RNA-seq response
The results from DE calculation from the combined data set were used to determine which genes were consistently up-regulated and down-regulated, respectively, across all clusters as a result of exposure to cocaine. The top 50 ranked differentially up-regulated genes for each cluster and the top 20 ranked differentially down-regulated genes for each cluster were input into the TopKLists R package (Schimek et al. 2015).
Cluster-specific coexpression networks
The scaled data from the sctransform pipeline for differentially expressed genes from clusters 16 and 22 were extracted for the male samples. These scaled data were used as input for filtering through the Random Matrix Theory (Gibson et al. 2013). The correlations that passed the filtering process were visualized using Cytoscape version 3.7.2. The MCODE algorithm (Bader and Hogue 2003) was utilized to identify highly interconnected modules within the larger cluster network. Genetic interaction networks were constructed by converting the gene IDs to gene names/symbols using the FlyBase Consortium's ‘Query-by-symbols/ID’ tool and calculating interactions between gene products using the StringApp plugin within Cytoscape (Doncheva et al. 2019). To identify specific pathways that are enriched in genes within each of the circular groups, we performed statistical overrepresentation tests on the gene IDs from each group using PANTHER (Thomas et al. 2003) and Reactome (Fabregat et al. 2018) databases. Pathways with BH-FDR adjusted P-values < 0.05 were considered statistically enriched.
Data access
All single-cell RNA sequences data generated in this study have been submitted to the NCBI Gene Expression Omnibus (GEO; https://www.ncbi.nlm.nih.gov/geo/) under accession number GSE152495. R code that was used to perform Seurat-based analyses with TopKLists is included in Supplemental Code and is available at GitHub (https://github.com/vshanka23/The-Drosophila-Brain-on-Cocaine-at-Single-Cell-Resolution/blob/master/Rcode_for_analyses.R).
Competing interest statement
The authors declare no competing interests.
Acknowledgments
We thank Marion Campbell for technical assistance. This work was supported by a grant from the National Institute on Drug Abuse (U01 DA041613) to T.F.C.M. and R.R.H.A.
Notes
[1] Supplementary material [Supplemental material is available for this article.]
[2] Article published online before print. Article, supplemental material, and publication date are at https://www.genome.org/cgi/doi/10.1101/gr.268037.120.
[3] Freely available online through the Genome Research Open Access option.
References
- ↵Bader GD, Hogue CW. 2003. An automated method for finding molecular complexes in large protein interaction networks. BMC Bioinformatics 4: 2. 10.1186/1471-2105-4-2
- ↵Badisa RB, Goodman CB, Fitch-Pye CA. 2013. Attenuating effect of N-acetyl-L-cysteine against acute cocaine toxicity in rat C6 astroglial cells. Int J Mol Med 32: 497–502. 10.3892/ijmm.2013.1391
- ↵Badisa RB, Fitch-Pye CA, Agharahimi M, Palm DE, Latinwo LM, Goodman CB. 2014. Milk thistle seed extract protects rat C6 astroglial cells from acute cocaine toxicity. Mol Med Rep 10: 2287–2292. 10.3892/mmr.2014.2524
- ↵Badisa RB, Kumar SS, Mazzio E, Haughbrook RD, Allen JR, Davidson MW, Fitch-Pye CA, Goodman CB. 2015. N-acetyl cysteine mitigates the acute effects of cocaine-induced toxicity in astroglia-like cells. PLoS One 10: e0114285. 10.1371/journal.pone.0114285
- ↵Bainton RJ, Tsai LT, Singh CM, Moore MS, Neckameyer WS, Heberlein U. 2000. Dopamine modulates acute responses to cocaine, nicotine and ethanol in Drosophila. Curr Biol 10: 187–194. 10.1016/s0960-9822(00)00336-5
- ↵Bainton RJ, Tsai LT, Schwabe T, DeSalvo M, Gaul U, Heberlein U. 2005. Moody encodes two GPCRs that regulate cocaine behaviors and blood-brain barrier permeability in Drosophila. Cell 123: 145–156. 10.1016/j.cell.2005.07.029
- ↵Becht E, McInnes L, Healy J, Dutertre C, Kwok IWH, Ng LG, Ginhoux F, Newell EW. 2019. Dimensionality reduction for visualizing single-cell data using UMAP. Nat Biotechnol 37: 38–44. 10.1038/nbt.4314
- ↵Becker JB, Koob GF. 2016. Sex differences in animal models: focus on addiction. Pharmacol Rev 68: 242–263. 10.1124/pr.115.011163
- ↵Borue X, Cooper S, Hirsh J, Condron B, Venton BJ. 2009. Quantitative evaluation of serotonin release and clearance in Drosophila. J Neurosci Methods 179: 300–308. 10.1016/j.jneumeth.2009.02.013
- ↵Borue X, Condron B, Venton BJ. 2010. Both synthesis and reuptake are critical for replenishing the releasable serotonin pool in Drosophila. J Neurochem 113: 188–199. 10.1111/j.1471-4159.2010.06588.x
- ↵Butler A, Hoffman P, Smibert P, Papalexi E, Satija R. 2018. Integrating single-cell transcriptomic data across different conditions, technologies, and species. Nat Biotechnol 36: 411–420. 10.1038/nbt.4096
- ↵Cao J, Dorris DM, Meitzen J. 2018. Electrophysiological properties of medium spiny neurons in the nucleus accumbens core of prepubertal male and female Drd1a-tdTomato line 6 BAC transgenic mice. J Neurophysiol 120: 1712–1727. 10.1152/jn.00257.2018
- ↵Chang H, Grygoruk A, Brooks E, Ackerson LC, Maidment NT, Bainton RJ, Krantz DE. 2006. Overexpression of the Drosophila vesicular monoamine transporter increases motor activity and courtship but decreases the behavioral response to cocaine. Mol Psychiatry 11: 99–113. 10.1038/sj.mp.4001742
- ↵Cho S, Muthukumar AK, Stork T, Coutinho-Budd JC, Freeman MR. 2018. Focal adhesion molecules regulate astrocyte morphology and glutamate transporters to suppress seizure-like behavior. Proc Natl Acad Sci USA 115: 11316–11321. 10.1073/pnas.1800830115
- ↵Claridge-Chang A, Wijnen H, Naef F, Boothroyd C, Rajewsky N, Young MW. 2001. Circadian regulation of gene expression systems in the Drosophila head. Neuron 32: 657–671. 10.1016/s0896-6273(01)00515-3
- ↵Corey YL, Quick MW, Davidson QN, Lester HA, Guastella J. 1994. A cocaine-sensitive Drosophila serotonin transporter: cloning, expression, and electrophysiological characterization. Proc Natl Acad Sci 91: 1188–1192. 10.1073/pnas.91.3.1188
- ↵Croset V, Treiber CD, Waddell S. 2018. Cellular diversity in the Drosophila midbrain revealed by single-cell transcriptomics. eLife 7: e34550. 10.7554/eLife.34550
- ↵Davie K, Janssens J, Koldere D, De Waegeneer M, Pech U, Kreft Ł, Aibar S, Makhzami S, Christiaens V, Bravo González-Blas C, 2018. A single-cell transcriptome atlas of the aging Drosophila brain. Cell 174: 982–998.e20. 10.1016/j.cell.2018.05.057
- ↵Demchyshyn LL, Pristupa ZB, Sugamori KS, Barker EL, Blakely RD, Wolfgang WJ, Forte MA, Niznik HB. 1994. Cloning, expression, and localization of a chloride-facilitated, cocaine-sensitive serotonin transporter from Drosophila melanogaster. Proc Natl Acad Sci 91: 5158–5162. 10.1073/pnas.91.11.5158
- ↵DeSalvo MK, Hindle SJ, Rusan ZM, Orng S, Eddison M, Halliwill K, Bainton RJ. 2014. The Drosophila surface glia transcriptome: evolutionary conserved blood-brain barrier processes. Front Neurosci 8: 346. 10.3389/fnins.2014.00346
- ↵Doncheva NT, Morris JH, Gorodkin J, Jensen LJ. 2019. Cytoscape StringApp: network analysis and visualization of proteomics data. J Proteome Res 18: 623–632. 10.1021/acs.jproteome.8b00702
- ↵Everett LJ, Huang W, Zhou S, Carbone MA, Lyman RF, Arya GH, Geisz MS, Ma J, Morgante F, Armour G, 2020. Gene expression networks in the Drosophila Genetic Reference Panel. Genome Res 30: 485–496. 10.1101/gr.257592.119
- ↵Fabregat A, Sidiropoulos K, Garapati P, Gillespie M, Hausmann K, Haw R, Jassal B, Jupe S, Korninger F, McKay S, 2016. The Reactome pathway Knowledgebase. Nucleic Acids Res 44(D1): D481–D487. 10.1093/nar/gkv1351
- ↵Fabregat A, Jupe S, Matthews L, Sidiropoulos K, Gillespie M, Garapati P, Haw R, Jassal B, Korninger F, May B, 2018. The Reactome Pathway Knowledgebase. Nucleic Acids Res 46(D1): D649–D655. 10.1093/nar/gkx1132
- ↵Filošević A, Al-Samarai S, Andretić Waldowski R. 2018. High throughput measurement of locomotor sensitization to volatilized cocaine in Drosophila melanogaster. Front Mol Neurosci 11: 25. 10.3389/fnmol.2018.00025
- ↵Ghasemzadeh MB, Vasudevan P, Giles C, Purgianto A, Seubert C, Mantsch JR. 2011. Glutamatergic plasticity in medial prefrontal cortex and ventral tegmental area following extended-access cocaine self-administration. Brain Res 1413: 60–71. 10.1016/j.brainres.2011.06.041
- ↵Gibson SM, Ficklin SP, Isaacson S, Luo F, Feltus FA, Smith MC. 2013. Massive-scale gene co-expression network construction and robustness testing using Random Matrix Theory. PLoS One 8: e55871. 10.1371/journal.pone.0055871
- ↵Greer CL, Grygoruk A, Patton DE, Ley B, Romero-Calderón R, Chang H, Houshyar R, Bainton RJ, Diantonio A, Krantz DE. 2005. A splice variant of the Drosophila vesicular monoamine transporter contains a conserved trafficking domain and functions in the storage of dopamine, serotonin, and octopamine. J Neurobiol 64: 239–258. 10.1002/neu.20146
- ↵Haas AL, Peters RH. 2000. Development of substance abuse problems among drug-involved offenders: evidence for the telescoping effect. J Subst Abuse 12: 241–253. 10.1016/S0899-3289(00)00053-5
- ↵Hafemeister C, Satija R. 2019. Normalization and variance stabilization of single-cell RNA-seq data using regularized negative binomial regression. Genome Biol 20: 296. 10.1186/s13059-019-1874-1
- ↵Harbison ST, Kumar S, Huang W, McCoy L, Smith KR, Mackay TFC. 2019. Genome-wide association study of circadian behavior in Drosophila melanogaster. Behav Genet 49: 60–82. 10.1007/s10519-018-9932-0
- ↵Haydon PG, Blendy J, Moss SJ, Jackson FR. 2009. Astrocytic control of synaptic transmission and plasticity: a target for drugs of abuse? Neuropharmacology 56(Suppl. 1): 83–90. 10.1016/j.neuropharm.2008.06.050
- ↵Highfill CA, Baker BM, Stevens SD, Anholt RRH, Mackay TFC. 2019. Genetics of cocaine and methamphetamine consumption and preference in Drosophila melanogaster. PLoS Genet 15: e1007834. 10.1371/journal.pgen.1007834
- ↵Hindle SJ, Bainton RJ. 2014. Barrier mechanisms in the Drosophila blood-brain barrier. Front Neurosci 8: 414. 10.3389/fnins.2014.00414
- ↵Holcroft CE, Jackson WD, Lin WH, Bassiri K, Baines RA, Phelan P. 2013. Innexins Ogre and Inx2 are required in glial cells for normal postembryonic development of the Drosophila central nervous system. J Cell Sci 126: 3823–3834. 10.1242/jcs.117994
- ↵Huang W, Massouras A, Inoue Y, Peiffer J, Ràmia M, Tarone AM, Turlapati L, Zichner T, Zhu D, Lyman RF, 2014. Natural variation in genome architecture among 205 Drosophila melanogaster Genetic Reference Panel lines. Genome Res 24: 1193–1208. 10.1101/gr.171546.113
- ↵Huang W, Campbell T, Carbone MA, Jones WE, Unselt D, Anholt RRH, Mackay TFC. 2020. Context-dependent genetic architecture of Drosophila life span. PLoS Biol 18: e3000645. 10.1371/journal.pbio.3000645
- ↵Ja WW, Carvalho GB, Mak EM, de la Rosa NN, Fang AY, Liong JC, Brummel T, Benzer S. 2007. Prandiology of Drosophila and the CAFE assay. Proc Natl Acad Sci 104: 8253–8256. 10.1073/pnas.0702726104
- ↵Jen JC, Wan J, Palos TP, Howard BD, Baloh RW. 2005. Mutation in the glutamate transporter EAAT1 causes episodic ataxia, hemiplegia, and seizures. Neurology 65: 529–534. 10.1212/01.wnl.0000172638.58172.5a
- ↵Jepson JE, Shahidullah M, Liu D, le Marchand SJ, Liu S, Wu MN, Levitan IB, Dalva MB, Koh K. 2014. Regulation of synaptic development and function by the Drosophila PDZ protein dyschronic. Development 141: 4548-4557. 10.1242/dev.109538
- ↵Kanda H, Shimamura R, Koizumi-Kitajima M, Okano H. 2019. Degradation of extracellular matrix by matrix metalloproteinase 2 is essential for the establishment of the blood-brain barrier in Drosophila. iScience 16: 218–229. 10.1016/j.isci.2019.05.027
- ↵Kariisa M, Scholl L, Wilson N, Seth P, Hoots B. 2019. Drug overdose deaths involving cocaine and psychostimulants with abuse potential—United States, 2003–2017. MMWR Morb Mortal Wkly Rep 68: 388–395. 10.15585/mmwr.mm6817a3
- ↵Kaun KR, Devineni AV, Heberlein U. 2012. Drosophila melanogaster as a model to study drug addiction. Hum Genet 131: 959–975. 10.1007/s00439-012-1146-6
- ↵Kounatidis I, Chtarbanova S. 2018. Role of glial immunity in lifespan determination: a Drosophila perspective. Front Immunol 9: 1362. 10.3389/fimmu.2018.01362
- ↵Kremer MC, Jung C, Batelli S, Rubin GM, Gaul U. 2017. The glia of the adult Drosophila nervous system. Glia 65: 606–638. 10.1002/glia.23115
- ↵Krzeptowski W, Walkowicz L, Płonczyńska A, Górska-Andrzejak J. 2018. Different levels of expression of the clock protein PER and the glial marker REPO in ensheathing and astrocyte-like glia of the distal medulla of Drosophila optic lobe. Front Physiol 9: 361. 10.3389/fphys.2018.00361
- ↵Levin LR, Han PL, Hwang PM, Feinstein PG, Davis RL, Reed RR. 1992. The Drosophila learning and memory gene rutabaga encodes a Ca2+ calmodulin-responsive adenylyl cyclase. Cell 68: 479–489. 10.1016/0092-8674(92)90185-f
- ↵Li H, Chaney S, Roberts IJ, Forte M, Hirsh J. 2000. Ectopic G-protein expression in dopamine and serotonin neurons blocks cocaine sensitization in Drosophila melanogaster. Curr Biol 10: 211–214. 10.1016/s0960-9822(00)00340-7
- ↵Little KY, Krolewski DM, Zhang L, Cassin BJ. 2003. Loss of striatal vesicular monoamine transporter protein (VMAT2) in human cocaine users. Am J Psychiatry 160: 47–55. 10.1176/appi.ajp.160.1.47
- ↵Liu LN, Zhu FP, Song MY, Kang XJ, Shang SJ, Zhang XY, Xu HD, Teng SS, Liu B, Kuo ST, 2014. Effect of cocaine on ion channels and glutamatergic EPSCs in noradrenergic locus coeruleus neurons. J Mol Neurosci 53: 345–351. 10.1007/s12031-013-0159-5
- ↵Lukas SE, Dorsey CM, Mello NK, Mendelson JH, Lundahl LH, Sholar M, Cunningham SL. 1996. Reversal of sleep disturbances in cocaine- and heroin-dependent men during chronic buprenorphine treatment. Exp Clin Psychopharmacol 4: 413–420. 10.1037/1064-1297.4.4.413
- ↵Mackay TFC, Richards S, Stone EA, Barbadilla A, Ayroles JF, Zhu D, Casillas S, Han Y, Magwire MM, Cridland JM, 2012. The Drosophila melanogaster Genetic Reference Panel. Nature 482: 173–178. 10.1038/nature10811
- ↵MacNamee SE, Liu KE, Gerhard S, Tran CT, Fetter RD, Cardona A, Tolbert LP, Oland LA. 2016. Astrocytic glutamate transport regulates a Drosophila CNS synapse that lacks astrocyte ensheathment. J Comp Neurol 524: 1979–1998. 10.1002/cne.24016
- ↵Matsuno M, Horiuchi J, Ofusa K, Masuda T, Saitoe M. 2019. Inhibiting glutamate activity during consolidation suppresses age-related long-term memory impairment in Drosophila. iScience 15: 55–65. 10.1016/j.isci.2019.04.014
- ↵Mazaud D, Kottler B, Gonçalves-Pimentel C, Proelss S, Tüchler N, Deneubourg C, Yuasa Y, Diebold C, Jungbluth H, Lai EC, 2019. Transcriptional regulation of the glutamate/GABA/glutamine cycle in adult glia controls motor activity and seizures in Drosophila. J Neurosci 39: 5269–5283. 10.1523/JNEUROSCI.1833-18.2019
- ↵McClung C, Hirsh J. 1998. Stereotypic behavioral responses to free-base cocaine and the development of behavioral sensitization in Drosophila. Curr Biol 8: 109–112. 10.1016/s0960-9822(98)70041-7
- ↵Moyer RA, Wang D, Papp AC, Smith RM, Duque L, Mash DC, Sadee W. 2011. Intronic polymorphisms affecting alternative splicing of human dopamine D2 receptor are associated with cocaine abuse. Neuropsychopharmacology 36: 753–762. 10.1038/npp.2010.208
- ↵Murakami S, Minami-Ohtsubo M, Nakato R, Shirahige K, Tabata T. 2017. Two components of aversive memory in Drosophila, anesthesia-sensitive and anesthesia-resistant memory, require distinct domains within the Rgk1 small GTPase. J Neurosci 37: 5496–5510. 10.1523/JNEUROSCI.3648-16.2017
- ↵Muriach M, López-Pedrajas R, Barcia JM, Sanchez-Villarejo MV, Almansa I, Romero FJ. 2010. Cocaine causes memory and learning impairments in rats: involvement of nuclear factor κ B and oxidative stress, and prevention by topiramate. J Neurochem 114: 675–684. 10.1111/j.1471-4159.2010.06794.x
- ↵Narendran R, Lopresti BJ, Mason NS, Deuitch L, Paris J, Himes ML, Kodavali CV, Nimgaonkar VL. 2014. Cocaine abuse in humans is not associated with increased microglial activation: an 18-kDa translocator protein positron emission tomography imaging study with [11C]PBR28. J Neurosci 34: 9945–9950. 10.1523/JNEUROSCI.0928-14.2014
- ↵Noble EP, Blum K, Khalsa ME, Ritchie T, Montgomery A, Wood RC, Fitch RJ, Ozkaragoz T, Sheridan PJ, Anglin MD, 1993. Allelic association of the D2 dopamine receptor gene with cocaine dependence. Drug Alcohol Depend 33: 271–285. 10.1016/0376-8716(93)90113-5
- ↵Norga KK, Gurganus MC, Dilda CL, Yamamoto A, Lyman RF, Patel PH, Rubin GM, Hoskins RA, Mackay TFC, Bellen HJ. 2003. Quantitative analysis of bristle number in Drosophila mutants identifies genes involved in neural development. Curr Biol 13: 1388–1396. 10.1016/s0960-9822(03)00546-3
- ↵Northcutt AL, Hutchinson MR, Wang X, Baratta MV, Hiranita T, Cochran TA, Pomrenze MB, Galer EL, Kopajtic TA, Li CM, 2015. DAT isn't all that: cocaine reward and reinforcement require Toll-like receptor 4 signaling. Mol Psychiatry 20: 1525–1537. 10.1038/mp.2014.177
- ↵Nuzhdin SV, Pasyukova EG, Dilda CL, Zeng ZB, Mackay TFC. 1997. Sex-specific quantitative trait loci affecting longevity in Drosophila melanogaster. Proc Natl Acad Sci 94: 9734–9739. 10.1073/pnas.94.18.9734
- ↵Pandey UB, Nichols CD. 2011. Human disease models in Drosophila melanogaster and the role of the fly in therapeutic drug discovery. Pharmacol Rev 63: 411–436. 10.1124/pr.110.003293
- ↵Parinejad N, Peco E, Ferreira T, Stacey SM, van Meyel DJ. 2016. Disruption of an EAAT-mediated chloride channel in a Drosophila model of ataxia. J. Neurosci 36: 7640–7647. 10.1523/jneurosci.0197-16.2016
- ↵Persico AM, Bird G, Gabbay FH, Uhl GR. 1996. D2 dopamine receptor gene taqI A1 and B1 restriction fragment length polymorphisms: enhanced frequencies in psychostimulant-preferring polysubstance abusers. Biol Psychiatry 40: 776–784. 10.1016/0006-3223(95)00483-1
- ↵R Core Team. 2013. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. http://www.R-project.org/.
- ↵Rodan AR, Kiger JAJr, Heberlein U. 2002. Functional dissection of neuroanatomical loci regulating ethanol sensitivity in Drosophila. J Neurosci 22: 9490–9501. 10.1523/jneurosci.22-21-09490.2002
- ↵Russo SJ, Wilkinson MB, Mazei-Robison MS, Dietz DM, Maze I, Krishnan V, Renthal W, Graham A, Birnbaum SG, Green TA, 2009. Nuclear factor κ B signaling regulates neuronal morphology and cocaine reward. J Neurosci 29: 3529–3537. 10.1523/jneurosci.6173-08.2009
- ↵Schimek MG, Budinská E, Kugler KG, Švendová V, Ding J, Lin S. 2015. TopKLists: a comprehensive R package for statistical inference, stochastic aggregation, and visualization of multiple omics ranked lists. Stat Appl Genet Mol Biol 14: 311–316. 10.1515/sagmb-2014-0093
- ↵Scholz N, Ehmann N, Sachidanandan D, Imig C, Cooper BH, Jahn O, Reim K, Brose N, Meyer J, Lamberty M, 2019. Complexin cooperates with Bruchpilot to tether synaptic vesicles to the active zone cytomatrix. J Cell Biol 218: 1011–1026. 10.1083/jcb.201806155
- ↵Shin CB, Serchia MM, Shahin JR, Ruppert-Majer MA, Kippin TE, Szumlinski KK. 2016. Incubation of cocaine-craving relates to glutamate over-flow within ventromedial prefrontal cortex. Neuropharmacology 102: 103–110. 10.1016/j.neuropharm.2015.10.038
- ↵Shorter J, Couch C, Huang W, Carbone MA, Peiffer J, Anholt RRH, Mackay TFC. 2015. Genetic architecture of natural variation in Drosophila melanogaster aggressive behavior. Proc Natl Acad Sci 112: E3555–E3563. 10.1073/pnas.1510104112
- ↵Simon AF, Daniels R, Romero-Calderón R, Grygoruk A, Chang HY, Najibi R, Shamouelian D, Salazar E, Solomon M, Ackerson LC, 2009. Drosophila vesicular monoamine transporter mutants can adapt to reduced or eliminated vesicular stores of dopamine and serotonin. Genetics 181: 525–541. 10.1534/genetics.108.094110
- ↵Sivachenko A, Li Y, Abruzzi KC, Rosbash M. 2013. The transcription factor Mef2 links the Drosophila core clock to Fas2, neuronal morphology, and circadian behavior. Neuron 79: 281–292. 10.1016/j.neuron.2013.05.015
- ↵Spéder P, Brand AH. 2014. Gap junction proteins in the blood-brain barrier control nutrient-dependent reactivation of Drosophila neural stem cells. Dev Cell 30: 309–321. 10.1016/j.devcel.2014.05.021
- ↵Stolf AR, Cupertino RB, Müller D, Sanvicente-Vieira B, Roman T, Vitola ES, Grevet EH, von Diemen L, Kessler FHP, Grassi-Oliveira R, 2019. Effects of DRD2 splicing-regulatory polymorphism and DRD4 48 bp VNTR on crack cocaine addiction. J Neural Transm (Vienna) 126: 193–199. 10.1007/s00702-018-1946-5
- ↵Substance Abuse and Mental Health Services Administration. 2019. Key substance use and mental health indicators in the United States: results from the 2018 National Survey on Drug Use and Health (HHS Publication No. PEP19-5068, NSDUH Series H-54). Center for Behavioral Health Statistics and Quality, Substance Abuse and Mental Health Services Administration, Rockville, MD. https://www.samhsa.gov/data/.
- ↵Thomas PD, Campbell MJ, Kejariwal A, Mi H, Karlak B, Daverman R, Diemer K, Muruganujan A, Narechania A. 2003. PANTHER: a library of protein families and subfamilies indexed by function. Genome Res 13: 2129–2141. 10.1101/gr.772403
- ↵Thurmond J, Goodman JL, Strelets VB, Attrill H, Gramates LS, Marygold SJ, Matthews BB, Millburn G, Antonazzo G, Trovisco V, 2019. FlyBase 2.0: the next generation. Nucleic Acids Res 47(D1): D759–D765. 10.1093/nar/gky1003
- ↵Tsacopoulos M, Magistretti PJ. 1996. Metabolic coupling between glia and neurons. J Neurosci 16: 877–885. 10.1523/jneurosci.16-03-00877.1996
- ↵Wang KH, Penmatsa A, Gouaux E. 2015. Neurotransmitter and psychostimulant recognition by the dopamine transporter. Nature 521: 322–327. 10.1038/nature14431
- ↵Westermeyer J, Boedicker AE. 2000. Course, severity, and treatment of substance abuse among women versus men. Am J Drug Alcohol Abuse 26: 523–535. 10.1081/ada-100101893
- ↵Wu X, Gu HH. 2003. Cocaine affinity decreased by mutations of aromatic residue phenylalanine 105 in the transmembrane domain 2 of dopamine transporter. Mol Pharmacol 63: 653–658. 10.1124/mol.63.3.653
- ↵Xu S, Chan T, Shah V, Zhang S, Pletcher SD, Roman G. 2012. The propensity for consuming ethanol in Drosophila requires rutabaga adenylyl cyclase expression within mushroom body neurons. Genes Brain Behav 11: 727–739. 10.1111/j.1601-183X.2012.00810.x
- ↵Yao L, Fan P, Arolfo M, Jiang Z, Olive MF, Zablocki J, Sun HL, Chu N, Lee J, Kim HY, 2010. Inhibition of aldehyde dehydrogenase-2 suppresses cocaine seeking by generating THP, a cocaine use-dependent inhibitor of dopamine synthesis. Nat Med 16: 1024–1028. 10.1038/nm.2200
- ↵Ye Y, Xi W, Peng Y, Wang Y, Guo A. 2004. Long-term but not short-term blockade of dopamine release in Drosophila impairs orientation during flight in a visual attention paradigm. Eur J Neurosci 20: 1001–1007. 10.1111/j.1460-9568.2004.03575.x
- ↵Zhang C, Ding H, Cheng Y, Chen W, Li Q, Li Q, Dai R, Luo M. 2017. Genetic polymorphisms in ALDH2 are associated with drug addiction in a Chinese Han population. Oncotarget 8: 8597–8603. 10.18632/oncotarget.14354
- ↵Zhu R, Bu Q, Fu D, Shao X, Jiang L, Guo W, Chen B, Liu B, Hu Z, Tian J, 2018. Toll-like receptor 3 modulates the behavioral effects of cocaine in mice. J Neuroinflammation 15: 93. 10.1186/s12974-018-1130-8
- ↵Zwarts L, Vanden Broeck L, Cappuyns E, Ayroles JF, Magwire MM, Vulsteke V, Clements J, Mackay TFC, Callaerts P. 2015. The genetic basis of natural variation in mushroom body size in Drosophila melanogaster. Nat Commun 6: 10115. 10.1038/ncomms10115

