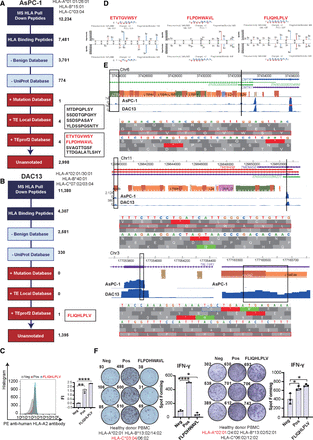
TE-chimeric transcript–derived peptides as neoantigens. (A) Annotation of 12,234 HLA-bound peptides identified by mass spectrometry (MS) from AsPC-1 cells across multiple reference databases, including benign, UniProt, mutation, locally translated TE ORFs, TEprof2, and unannotated regions. (B) Annotation of 11,380 HLA-bound peptides identified from DAC13 organoids, with one TEprof2-derived peptide (FLIQHLPLV) retained for further analysis. (C) Validation of the binding affinity of the TEprof2-derived peptide FLIQHLPLV to HLA-A*02:01 using T2 binding assay. (D) Spectral comparison between MS-identified endogenous peptides and synthetic reference peptides: ETVTGVWSY and FLPDHWAVL (AsPC-1), and FLIQHLPLV (DAC13), confirming accurate peptide identification. (E) Visualization of TE-chimeric transcripts and translation evidence using the WashU Epigenome Browser. TE loci are shown with RNA-seq read coverage and getorf-based predicted ORFs; corresponding peptide-coding regions are highlighted. (F) Immunogenicity validation of TE-derived peptides (FLPDHWAVL and FLIQHLPLV) in ELISPOT assays using HLA-genotyped healthy donor PBMCs. Significant IFNG responses indicate these peptides elicit T cell activation.











