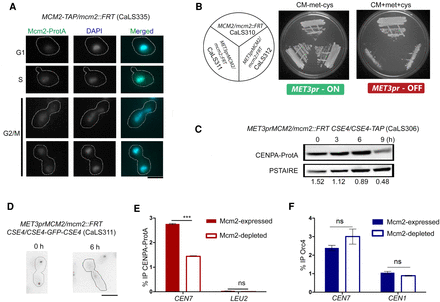
Mcm2 is essential for cell viability and CENPA stability in C. albicans. (A) Intracellular localization of Mcm2–Protein A in CaLS335 (MCM2-TAP/mcm2::FRT) cells stained with anti–Protein A antibodies and DAPI. Scale bar, 5 µm. (B) CaLS310 (MCM2/mcm2::FRT), where one copy of MCM2 was deleted, and two independent transformants, CaLS311 and CaLS312 (MET3prMCM2/mcm2::FRT), where the remaining wild-type copy was placed under the control of the MET3 promoter, were streaked on plates containing permissive (CM-met-cys) and nonpermissive (CM + 5 mM cys + 5 mM met) media and photographed after 48 h of incubation at 30°C. (C) Western blot of the whole-cell lysate of CaLS306 (MET3prMCM2/mcm2::FRT CSE4/CSE4-TAP) using anti–Protein A antibodies revealed a time-dependent decrease in CENPA levels upon depletion of Mcm2 for 3, 6, and 9 h. Normalization was performed using PSTAIRE. (D) CENPA (GFP) cluster delocalizes upon depletion of Mcm2 in CaLS311 (MET3prMCM2/mcm2::FRT CSE4-GFP-CSE4/CSE4). Scale bar, 5 µm. n = 100. (E) ChIP-qPCR using anti–Protein A antibodies revealed a significant reduction of CENPA at CEN7 in CaLS306 when grown in nonpermissive media for 6 h. Two-way ANOVA; (***) P-value < 0.001; (ns) P-value > 0.05; n = 3. (F) ChIP-qPCR in CaLS311 revealed no significant difference in the Orc4 enrichment at the centromeres in the presence or absence of Mcm2. Two-way ANOVA; (***) P-value < 0.001; (ns) P-value > 0.05; n = 3.











