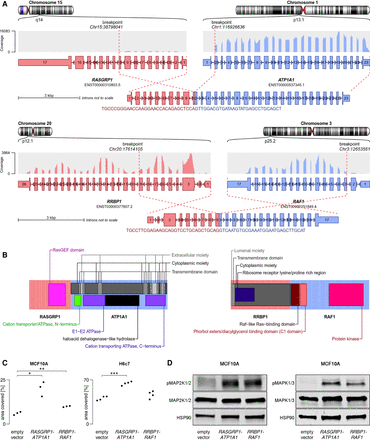
Structural and functional characteristics of RRBP1-RAF1 and RASGRP1-ATP1A1. (A) Structure of the fusion transcripts. (B) Protein domains retained in the fusion proteins and topology. Near full-length RAF1 was found to be fused to the transmembrane protein RRBP1, presumably tethering RAF1 to the endoplasmatic reticulum with its kinase domain facing the cytoplasmic space. The oncogene RASGRP1 was predicted to be fused to ATP1A1, a protein embedded in the plasma membrane. Although oncogenes are more often found to constitute the C terminus of a fusion protein, RASGRP1 appeared to be fused to the N terminus of ATP1A1, thereby replacing several C-terminal domains of RASGRP1, which normally regulate recruitment to the plasma membrane, where RASGRP1 activates its target, KRAS (Beaulieu et al. 2007). Presumably, replacement of these regulatory domains by a membrane-bound protein increased the activity of RASGRP1 by means of warranting proximity to KRAS. (C) MCF10A and H6c7 cells were stably transduced with one of the fusion constructs or empty vector. MCF10A cells were cultured for 8 d without EGF, H6c7 cells were cultured for 7 d with EGF, and the area covered by cells was measured. Statistical significance was tested using a two-sided Welch t-test (MCF10A RASGRP1-ATP1A1: P-value = 0.023; MCF10A RRBP1-RAF1: P-value = 0.0094; H6c7 RASGRP1-ATP1A1: P-value = 4.1×10−5; H6c7 RRBP1-RAF1: P-value = 0.14). (D) Western blot showing increased phosphorylation of MAP2K1/2 (MEK1/2) and MAPK1/3 (ERK2/1) in TP53-deficient MCF10A cells stably transduced with one of the fusions as compared to empty vector.











