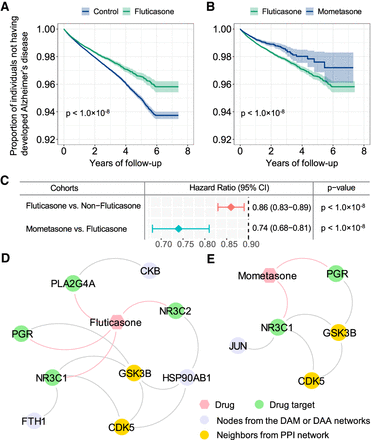
Retrospective case-control analysis reveals that usage of fluticasone and mometasone is significantly associated with reduced likelihood of AD in a longitudinal patient database with 7.23 million subjects. Two comparison analyses were conducted: (A) fluticasone (a glucocorticoid receptor agonist) versus a matched control population (non-fluticasone users), and (B) mometasone (a stronger glucocorticoid receptor agonist) versus fluticasone. For each comparator, we estimated the unstratified Kaplan–Meier curves, conducted propensity score–stratified (n strata = 10) rank test and applied Cox models after adjusting all possible confounding factors, including age, gender, race, and disease comorbidities (Supplemental Table S16). (C) Hazard ratios (HRs) and 95% confidence interval (CI) for two drug cohort studies. Propensity score–stratified Cox-proportional hazards models were used to conduct statistical inference for the hazard ratios. (D,E) Proposed mechanism-of-action for treatment of AD by fluticasone and mometasone using drug-target network analysis.











