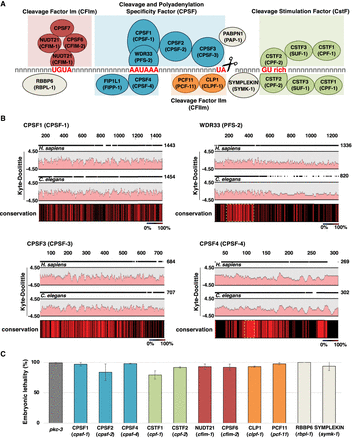
The C. elegans members of the cleavage and polyadenylation complex (CPC). (A) The CPC is composed of at least four independent subcomplexes named cleavage and polyadenylation specificity complex (blue), which canonically recognizes the PAS hexamer “AAUAAA” and performs the cleavage downstream from the dinucleotide TA; the cleavage stimulation factor complex (green), which binds downstream from the cleavage site to GU-rich elements; and the cleavage factor CFIm (red) and CFIIm (orange) complexes. CFIm recognizes the element “UGUA” located upstream of the PAS element. This element is not always present. Other known required factors are the poly(A) polymerase enzyme, the scaffolding member symplekin, and RBBP6. The names of the C. elegans orthologs are shown in parentheses. (B) The human and C. elegans CPSF subcomplexes are similar in amino acid composition and structure. Two-species alignments between several members of the human and C. elegans CPSF members. Amino acids 100% conserved between these two species are shown in red in the conservation bar. Yellow dashed boxes show the sequence of the proteins that interact with the PAS element. Functional domains are conserved. The two Kyte-Doolittle graphs in each panel indicate the hydrophobic amino acids in human and C. elegans. (C) RNAi was used to selectively silence most of the members of the CPC complex in C. elegans. We observed a strong embryonic lethality phenotype with all the RNAi experiments performed.











