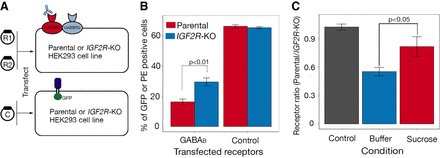
IGF2R traffics GABBR2 through a clathrin-dependent pathway. (A) Schematic of a flow cytometry–based GABAB cell surface expression assay. Parental and IGF2R-KO HEK293 cells were cotransfected with plasmids encoding both GABBR1 (R1) and GABBR2 (R2) full-length cDNAs and surface expression of GABAB receptors quantified by antibody staining and FACS. A plasmid containing a GFP-tagged CD200 cDNA was transfected in parallel and the number of transfected cells quantified by GFP fluorescence. (B) The percentage of GABAB receptor–positive cells was significantly higher on the surface of IGF2R-KO cells compared with the parental line (P < 0.01 using Welch's two sample t-test, n = 3); control demonstrates both cell lines were transfected at comparable levels. Error bars, ± SD, n = 3 (cells were transfected in triplicate); a representative experiment of two independent experiments is shown. (C) The clathrin-pathway inhibitor sucrose was added to parental and IGF2R-deficient HEK293 cells expressing the GABAB receptor complex, and the cell surface receptor levels were quantified. The effect of sucrose on the IGF2R–GABAB receptor complex interaction was determined by calculating the ratio of positively stained cells on the parental cell line relative to the IGF2R-deficient cells and a buffer-only control. Sucrose treatment led to a significant increase (P < 0.05 using Welch's two sample t-test) in cell surface GABAB receptor complex staining on the parental cell line, almost to the same level as on the IGF2R-deficient cells, demonstrating that that GABAB receptor trafficking depends on the clathrin pathway. The control represents a transfection control to demonstrate parental and IGF2R-deficient cells were transfected with equal efficiency. Bars, mean ± SD; n = 3 (cells were transfected in triplicate).











