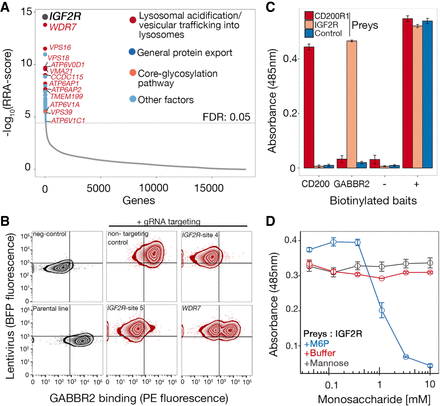
IGF2R interacts with GABBR2 in a mannose-6-phosphate (M6P)–dependent manner. (A) RRA-score rank-ordered genes identified from gRNA enrichment analysis from sorted mutant cells that had lost GABBR2 binding activity. Enriched genes encoded the IGF2R receptor and proteins involved in lysosome biology (also see Supplemental Data S3). (B) Binding of GABBR2 was quantified on HEK293 cells transduced with two gRNAs targeting different exons of IGF2R and one gRNA targeting WDR7. A near complete loss of binding was observed on IGF2R-targeted cells and a partial loss on WDR7-targeted cells; targeted cells were maintained as polyclonal lines. A representative experiment of three technical replicates is shown. (C) Direct binding between IGF2R and GABBR2 ectodomains. The biotinylated ectodomain of GABBR2 was immobilized as a “bait” on streptavidin-coated microtiter plates and tested for direct interactions using a beta-lactamase–tagged “prey” ectodomain of IGF2R. Binding was quantified using the beta-lactamase substrate nitrocefin, whose hydrolysis products absorb at 485 nm. Positive control was the CD200–CD200R1 interaction; control “prey” is an unrelated ectodomain, positive (+) represents total capture of all preys with an anti-prey antibody, and negative (−) represents a tag-only bait control. (D) The interaction between IGF2R and GABBR2 can be completely inhibited by 10 mM soluble M6P. The binding dependency on M6P was tested by adding serial dilutions of mannose, M6P, or buffer alone. Data points in C and D are mean ± SEM, n = 3.











