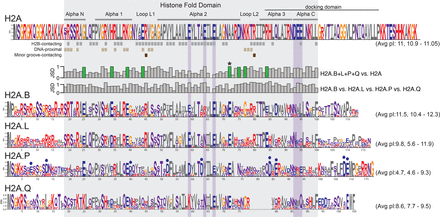
Short H2A variants have distinct protein features. Logo plots of protein alignments of canonical and short H2As across an identical set of representative eutherian mammals (except for H2A.Q) (see Methods). Residues are colored based on biochemical proprieties; hydrophobic residues in gray; positively charged in red; negatively charged in blue; polar in purple, and others (Gly and Cys) in orange. Residues in proximity to H2B (gray squares, at least 4 Å), DNA (light brown, 5 Å), or buried in the minor groove (dark brown) are annotated below the H2A logo. The histone fold domain and the six dispersed residues of the acidic patch are boxed in gray and purple, respectively. Jensen-Shannon distances (JSD) measured at each amino acid position between combinations of logos are indicated as bar graphs (low values meaning very similar, high values meaning very different). Charge-altering changes that are conserved among short H2As but different from canonical H2A are highlighted using green-colored bars. Residues uniquely altered to acidic residues in H2A.P are highlighted using blue dots. The average and range of isoelectric points (pI) across the selected mammals are shown in parentheses.











