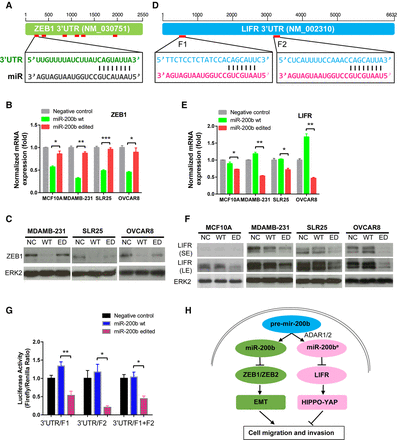
Molecular mechanisms of WT and edited miR-200b in cancer cells. (A) 3′ UTR representation of WT miR-200b target gene ZEB1. (B) qRT-PCR of ZEB1 upon 24-h transfection with WT miR-200b and edited miR-200b mimics in MCF10A, MDAMB-231, SLR25, and OVCAR8 cells. (C) Western blots of ZEB1 upon 48-h transfection with WT miR-200b and edited miR-200b mimics in MDAMB-231, SLR25, and OVCAR8 cells. (D) 3′ UTR representation of edited miR-200b target, LIFR. (E) qRT-PCR of LIFR upon 24-h transfection with WT miR-200b and edited miR-200b mimics in MCF10A, MDAMB-231, SLR25, and OVCAR8 cells. (F) Western blots of LIFR upon 48-h transfection with WT miR-200b and edited miR-200b mimics in MCF10A, MDAMB-231, SLR25, and OVCAR8 cells. Blots with short-time (SE) and long-time exposure (LE) are shown. (G) Luciferase reporter assays that contain two predicted binding sites of edited miR-200b (F1 and F2) in LIFR. In B, E, and G, two-sided t-test was used to assess the difference, n = 2 or 3, and error bars denote ±SEM; (*) P < 0.05. (**) P < 0.01, (***) P < 0.001. (H) Proposed mechanistic model in which WT miR-200b inhibits key EMT regulators ZEB1 and ZEB2, thereby suppressing cell migration and invasion, whereas edited miR-200b (catalyzed by both ADAR1 and ADAR2) inhibits a new target LIFR, a well-characterized metastasis suppressor, thereby promoting cell migration and invasion.











