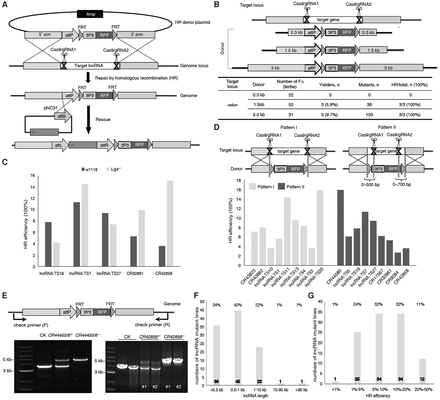
CRISPR/Cas9-mediated high-throughput mutagenesis of 105 lncRNAs. (A) Schematic representation of the strategy for generating lncRNA knockout and rescue lines using CRISPR. (B) Determination of the optimal HR donor length for efficient gene replacement. The plasmid containing a reporter cassette (attP-FRT-3P3-RFP) flanked by 0.3, 1.5, and 3 kb HR sequences was provided as the donor DNA. The oskar gene was used to test the system. (C) DNA Ligase4 deficiency had no dramatic effect on HR efficiency at different target loci. (D) Within a range of 0–700 bp, the distance between the double-strand break site and the homology arm had no dramatic effect on HR frequency. (E) Genotyping of w1118, lncRNA+/−, and lncRNA−/− flies to confirm lncRNA knockouts. Insertion of the reporter cassette results in a size increase in the PCR products, which is visualized only in heterozygous and homozygous flies. (F) Length distribution of the targeted lncRNAs. (G) The distribution of HR efficiency when generating the 105 lncRNA mutants using CRISPR. F0 injected flies were crossed to w1118, and their progeny were screened for RFP-positive eyes. Crosses producing one or more transgenic progeny were considered as founder lines. The homologous recombination efficiency was calculated as the ratio between the number of founder lines and the number of F0 crosses.











