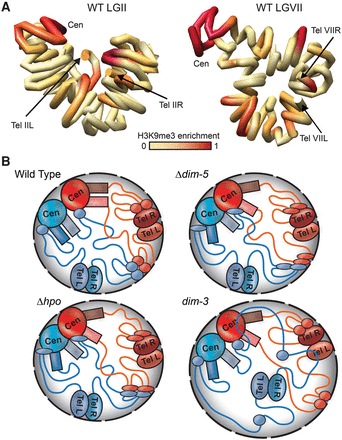
In silico chromosome modeling and schematics depicting genomic interactions in WT and mutant strains. (A) Computationally determined three-dimensional polymer models of Neurospora LGII (left) and LGVII (right) with chromosomes shown as continuous tubes. The path of each tube passes through the center of a series of 50-kb spheres, the relative conformation of which were determined using the contact frequencies in the WT Hi-C data set to set attractive or repulsive forces between each sphere and allowing the system to relax to a low energy state. The precise path of chromatin shown in these models may not be biologically relevant. Degree of WT H3K9me3 enrichment is shown in red. (B) Models for chromosome conformations in WT and heterochromatin-deficient strains for a nucleus with two chromosomes (blue and red), centromeric (CEN), pericentromeric (colored rectangles), subtelomeric (TEL), and interspersed heterochromatic (colored ovals) regions are indicated, and euchromatin is indicated as colored lines. (Top left) In a WT strain, inter- and intra-chromosomal heterochromatic regions bundle to dominate the genomic interaction landscape near the nuclear periphery (gray shaded area), with strong inter-centromeric as well as inter- and intra-chromosomal pericentromeric interactions. Although Hi-C contact data suggest subtelomeres do not frequently contact centromeres, in silico modeling (A; Supplemental Figs. S29, S30) suggests all heterochromatic regions may be in close proximity in the nucleus. (Top right) In a Δdim-5 strain, which is devoid of H3K9me3 and thus specific HP1 localization, the pericentromeric and subtelomeric regions have reduced interactions, whereas some centromeric and less compact interspersed heterochromatic regions (thin ovals) have increased contacts. (Bottom left) In a Δhpo strain, where heterochromatin retains H3K9me3 but has no HP1, the overall chromosome conformation is similar to Δdim-5. (Bottom right) In dim-3, which carries a mutated form of importin alpha that severely reduces H3K9me3 levels but retains near WT levels of HP1 (Klocko et al. 2015), telomere clusters (and some associated interspersed heterochromatic regions) become uncoupled from the nuclear periphery due to enlarged nuclear membrane diameter (black dashed outlines), thus increasing heterochromatic–euchromatic contacts and compromising the heterochromatic bundle.











