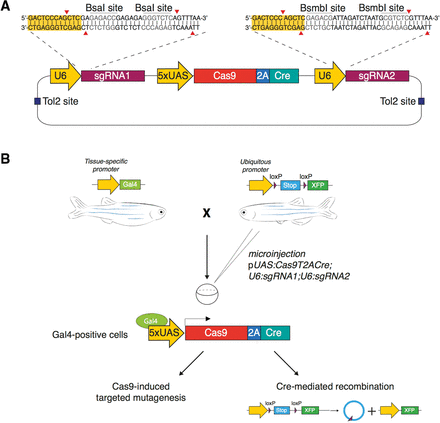
Permanent labeling of Cas9-expressing cells by Cre-mediated recombination. (A) Schematic illustration of the pUAS:Cas9T2ACre;U6sgRNA1;U6sgRNA2 expression vector design. The vector contains two U6-sgRNA expression cassettes targeting the same gene. BsaI and BsmBI restriction sites are used for the sgRNA target sequence cloning. UAS elements drive the expression of Cas9 and Cre recombinase linked via the T2A peptide. (B) Microinjection of the pUAS:Cas9T2ACre;U6sgRNA1;U6sgRNA2 construct in the double transgenic embryos Tg(Tissue specific promoter:Gal4) × Tg(Ubiquitous promoter:loxP-STOP-loxP-XFP) triggers simultaneous synthesis of Cas9 and Cre in a chosen spatiotemporal pattern, defined by the promoter driving Gal4 expression. Cell-specific Cas9 endonuclease activity induces targeted disruption of the gene of interest and Cre-mediated recombination induces permanent expression of a fluorescent reporter gene (XFP) by deletion of the floxed stop sequence.











