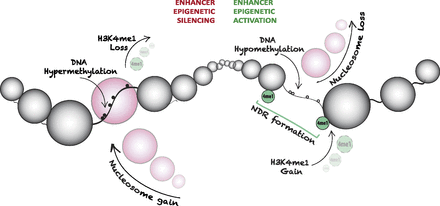
Model depicting mechanisms by which the distal regulatory architecture provides an additional layer of epigenetic plasticity in cancer. Enhancers preferentially undergo epigenetic silencing, which is exemplified by collapse of a NDR (nucleosome gain), DNA hypermethylation, and loss of the H3K4me1 mark. Epigenetic switching can also encompass aberrant epigenetic activation of enhancers. Here, an atypical NDR is formed due to nucleosome loss, and the enhancer becomes marked by H3K4me1, which is accompanied by DNA hypomethylation events in the cancer cells. Our data indicate that many more enhancers are abnormally silenced than activated, consistent with the hypothesis that it is more difficult to activate an already silenced regulatory element than it is to silence an already active or poised regulatory element. These findings also support a permanently silenced state in cancer. Thus, our data demonstrate that the global epigenetic landscape is dynamically altered at key regulatory regions outside of promoters. (NDR) Nucleosome-depleted region; (small white circle) unmethylated CpG site; (small black circle) methylated CpG site; (large circle) nucleosome; (4me1 [green]) H3K4me1.











