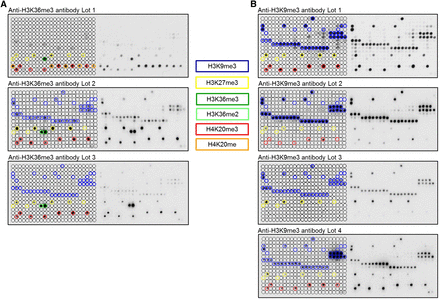
Peptide array analyses showing lot-to-lot fluctuations, cross-reactivity, and effects of proximal marks on the binding of popular histone tail antibodies. Peptide spots are annotated on the left side of the glass slide. The color-coded boxes denote the presence of the designated modifications. (A) Anti-H3K36me3 antibodies showed significant cross-reactivity to H4K20me1, H4K20me3, H3K27me3, H3K9me3, and H3K36me2. All lots of the H3K36me3 antibody had a weak binding specificity, and they showed very different properties. (B) Anti-H3K9me3 antibodies displayed cross-reactivity to H3K27me3 (Lot 3 even prefers H3K27me3 peptides) and H4K20me3 (very overt in the case of Lot 1 and Lot 4). H3K9me3 peptide spots that were not bound by these antibodies generally also contained H3S10ph or H3T11ph, indicating that this secondary mark prevents binding. For a more detailed annotation of the modifications at each spot, refer to Supplemental File S2. Among the H3K9me3 antibodies, only Lot 2 showed a binding specificity of acceptable quality. Anti-H3K36me3 (Lot 1) and anti-H3K9me3 (Lot 1) data were taken from Bock et al. (2011a) and reprocessed. Additional examples of commercial H3K36me3 antibodies with insufficient specificity are shown in Bock et al. (2011a). Examples of the fundamental differences in the specificity profiles of different antibodies directed against H3K27me3 are given in Supplemental Figure S1.











