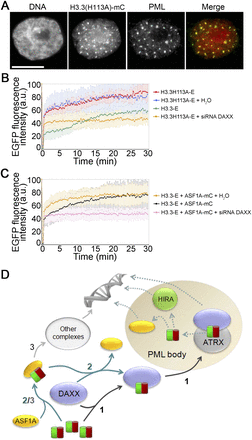
Accelerated recruitment of an (H3.3–H4)2 tetramerization mutant to PML bodies. (A) Colocalization of an H3.3(H113A)-mC tetramerization mutant with immunolabeled PML, 24 h after transfection. (B) FRAP analysis of H3.3(H113A)-EGFP in NBs with and without siRNA-mediated down-regulation of DAXX. H3.3-EGFP recovery was examined in parallel as control. H3.3(H113A)-EGFP displays a greater soluble pool than H3.3-EGFP, and recovery at NBs is abolished by DAXX knock-down (see Supplemental Fig. 7A for fluorescence recovery of the soluble fraction and at NBs) (mean±SD of 7-14 cells). (C) FRAP analysis of H3.3-EGFP in NBs in cells expressing ASF1A-mC with or without siRNA-mediated DAXX knock-down (mean±SD of 10-17 cells). (D) Model of (H3.3–H4) dimer recruitment to PML bodies prior to chromatin deposition. In pathway 1, DAXX recruits the (H3.3–H4) dimer to PML bodies. This process is facilitated by ASF1A (pathway 2). (H3.3–H4) loaded onto ASF1A can also be brought to additional H3.3 chaperone containing complexes (Szenker et al. 2011) (pathway 3). DAXX, ATRX, HIRA, and ASF1A are localized in a proportion of PML bodies and may speculatively be available for loading (H3.3–H4) prior to deposition into chromatin (dashed arrows).











