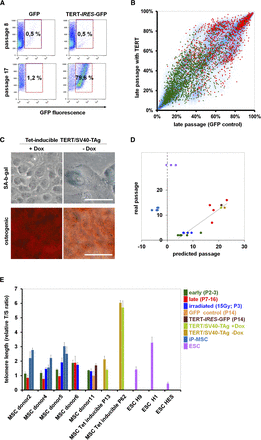
Immortalization does not affect SA-DNAm. (A) MSC at passage 2 were either retrovirally transfected with an TERT-IRES-GFP construct or with IRES-GFP (control; n = 5). Flow cytometic analysis of one sample reveals a growth advantage of TERT-transduced cells at late passages. (B) The DNAm profile of this TERT-transfected sample was compared with the corresponding control (both passage 14). CpG sites that were hypermethylated (red) or hypomethylated upon long-term culture (green) are indicated. Overall, TERT expression does not affect SA-DNAm changes. (C) MSC were immortalized by tetracycline-inducible expression of TERT and SV40-TAg. Upon Dox removal from the culture medium, these cells acquire a typical senescent morphology within 10 d, express SA-β-gal, and lose osteogenic differentiation potential. (D) The Epigenetic-Senescence-Signature was used to estimate cellular aging of all samples (Koch et al. 2012). DNAm was determined at six SA-CpG sites by pyrosequencing and used for the linear regression model to predict the number of passages. The predictions correlate with the real passage number for all MSC samples, including irradiated, TERT-transfected, and TERT/SV40-TAg immortalized cells. In contrast, senescence-predictions for iP-MSC and ESC rather correspond to noncultured cells. (E) Relative telomere length was determined by the telomere to single-copy gene (T/S) ratio with quantitative multiplex PCR (each sample measured in triplicate). Telomere length is significantly reduced in MSC of later passage, whereas it is not affected by irradiation. TERT expression, TERT/SV40-TAg immortalization (particularly at passage 62), and generation of iP-MSC result in telomere extension.











