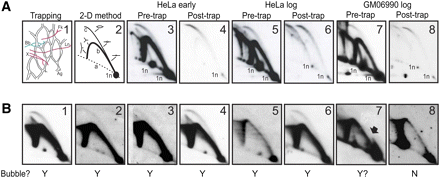
The trapped material, as well as the early S-phase HeLa test library, are extremely pure. (A) Frame 1 illustrates the principle of the bubble-trapping procedure, with agarose (Ag) indicated by the network, bubbles (Bb) indicated in red, and single forks (Fk), termination structures (X), and linear fragments (L) indicated in green. Frame 2 diagrams a 2D gel separation of replication intermediates, in which fragments with centered bubbles trace curve c, single forks trace curve b, and linear fragments correspond to the 1n spot. Frames 3, 4, 5, 6, 7, and 8 illustrate the composition of replication intermediates in the rDNA origins before and after trapping in agarose for the early S-phase HeLa sample, one of the log-phase HeLa samples, and the log-phase GM06990 sample, respectively. Note that all samples display two 1n spots, which arise from an RFLP at an EcoRI site in the multiple copies of the rDNA locus; only the smaller 12-kb RFLP is detected in the early S-phase samples, while 12- and 18-kb variants are both detected in the log-phase HeLa and GM06990 samples. (B) Eight anonymous cloned fragments from the early S-phase library ranging from 4 kb to 11 kb in length were tested for authenticity by analyzing their cognate genomic restriction fragments with suitable single-copy probes in 2D gels. Fragments 1–6 all display a composite pattern (complete bubble and single fork arc) indicative of initiation zones, while fragment 7 may correspond to a fixed, off-centered origin. The Y and N below each panel indicate whether each candidate actually displayed a bubble arc.











