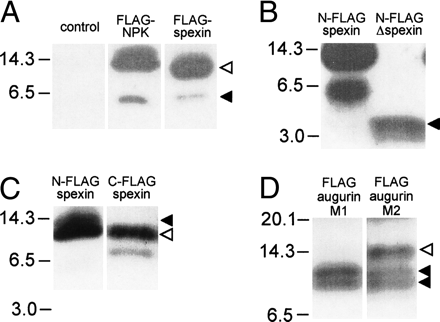
Identification of spexin and augurin in cell supernatants. Vector control and Flag-tagged NPK, spexin, and augurin were transfected into rat pancreatic cells in culture, and cell supernatants were harvested and submitted to immunoblotting with a Flag antibody. (A) Supernatants from Flag-NPK and N-Flag-spexin transfected cells contained high (solid arrow) and low (open arrow) mobility bands that reflected processing of Flag-tagged products from these constructs. (B) Supernatant from N-Flag-Δspexin transfected cells contained a 4-kDa band, suggesting that the 6-kDa product seen for N-Flag-spexin was the result of cleavage significantly C-terminal to the spexin peptide. (C) Supernatant from C-Flag-spexin contained two bands (12 and 8 kDa), confirming C-terminal cleavage of spexin pro-peptide. A 12-kDa product is seen for both N-Flag and C-Flag spexin (open arrow), while a 13-kDa product is seen only in N-Flag-spexin and corresponds to incompletely N-terminally processed spexin (closed arrow). (D) The presence of two bands (10 and 8 kDa, solid arrows) in supernatant from Flag-augurin transfected cells probed with M1 Flag antibody demonstrated cleavage of augurin at the putative pro-hormone cleavage site as well as close to the C terminus of the pro-peptide. The same immunoblot probed with M2 Flag antibody revealed an additional low-mobility product, confirming cleavage at the predicted dibasic cleavage site immediately adjacent to the Flag tag (open arrow).











