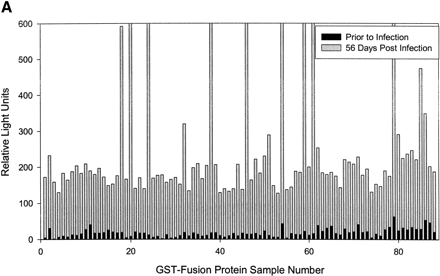
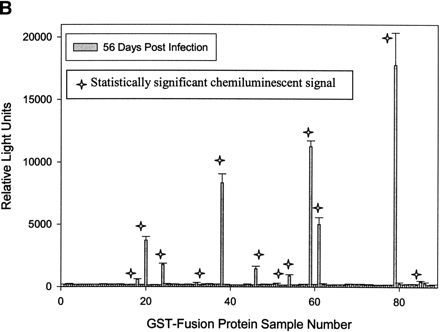
(A) Rapid identification of antigenic GST-fusion clones by a chemiluminescent immunoassay technique. Individual GST-fusion Treponema pallidum proteins from crude Escherichia coli extracts were selectively immobilized on glutathione-coated 96-well plates and were exposed to rabbit serum from rabbits either before or 2 months after intratesticular injection with virulent T. pallidum. Detection of binding interactions between rabbit IgG antibody and immobilized T. pallidum proteins was carried out with donkey, anti-rabbit IgG antibody conjugated to horseradish peroxidase. A plate reader monitored chemiluminescence, and the mean relative signal is presented for 85 T. pallidum fusion proteins, samples 3–87. Sample numbers 1 and 2 were prepared from E. coli that does not express a fusion protein. Sample 88 is the Borrelia burgdorferi fibronectin binding protein (BBK32) fused to GST expressed from pMM110-pUNI-D. (B) The mean relative signal and SD data for rabbit serum collected 2 months after intratesticular injection of T. pallidum is presented. A star has been placed above each of the 12 samples with a statistically significant signal.











