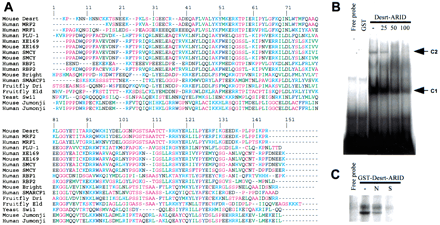
(A) The alignment of amino acids from ARID domains. Aromatic amino acids (F, Y, W) are dark blue; sulphydrylic amino acids (C) are red; basic amino acids (K, R, H) are light blue; aliphatic amino acids (V, I, L, M) are green; hydrophilic (1) amino acids (P, A, G, S, T) are pink; hydrophilic (2) amino acids (N, E, D, Q) are black. (B) Electrophoretic mobility shift assay (EMSA) of recombinant Desrt–ARID domain binding to a (TTA)9 oligonucleotide (lane 3). The main Desrt–-retarded complex is indicated by C1 and a lower mobility complex indicated by C2. Competition was performed with a 25-, 50-, or 100-fold excess of unlabeled oligonucleotide (lanes4–6). No binding was observed by use of GST alone (lane2) or in the absence of protein (lane 1, free probe). (C) EMSA of recombinant GST–Desrt–ARID fusion protein binding to a (TTA)9 oligonucleotide (lane 2). Competition was performed with a 100-fold excess of unlabeled nonspecific (N) oligonucleotide (lane 3) or specific (S) oligonucleotide (lane4). No binding was observed in the absence of protein (lane1, free probe).











