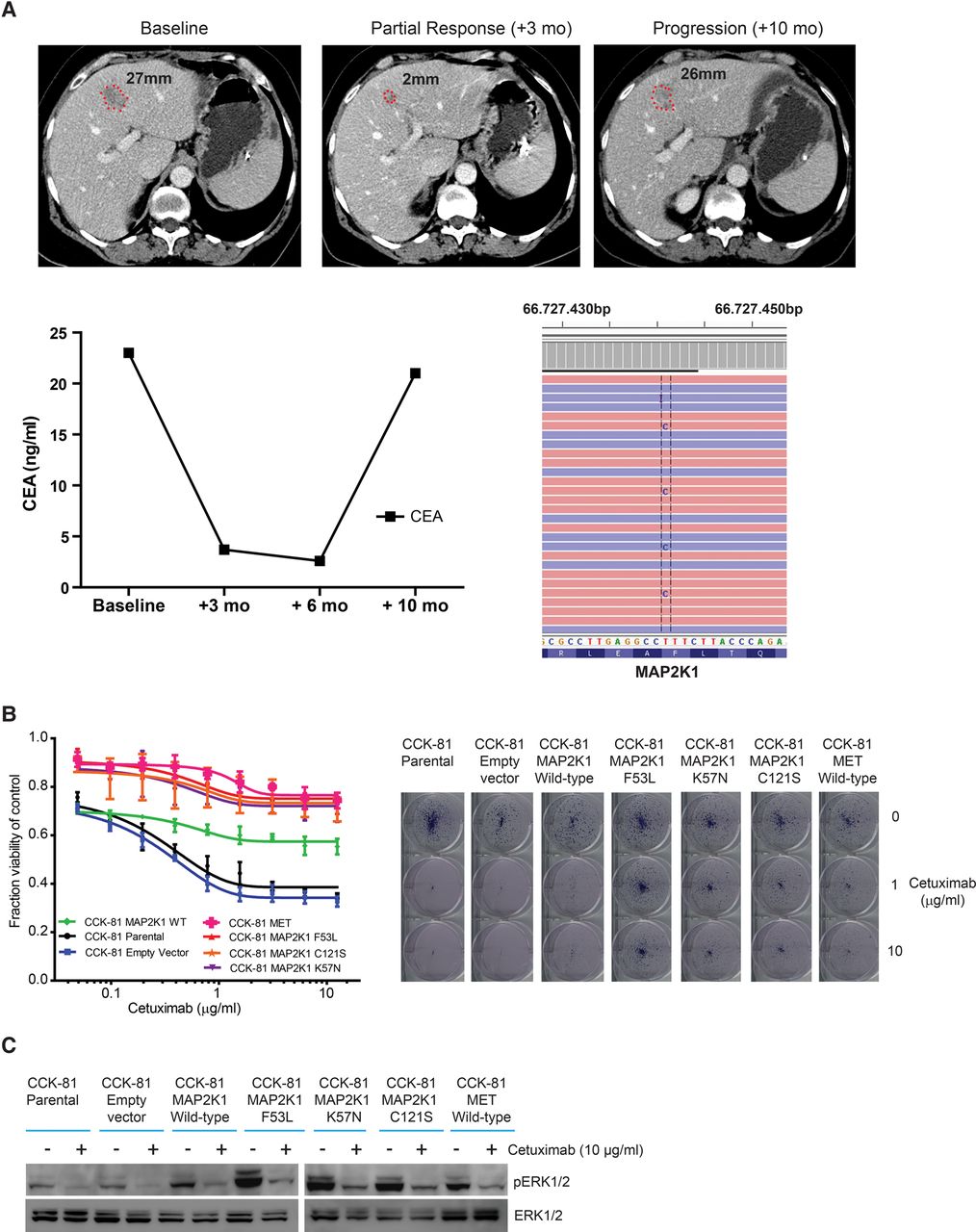
ENU mutagenesis identifies a novel MAP2K1 resistance mutation that is detected in a colorectal cancer patient after an initial response to Cetuximab. (A) A colorectal cancer patient with inoperable liver metastases was treated with the combination of FOLFIRI chemotherapy and Cetuximab. Initial response to treatment was demonstrated radiologically at 3 mo and by falling CEA marker levels. Subsequent measurements at 10 mo confirmed progressive disease, and sequencing of plasma DNA detected a novel mutation in MAP2K1, p.F53L. (B) CCK-81 cells were transduced with the following lentiviral MAP2K1 vectors: empty vector (mock), wild-type (WT), K57N, C121S, and F53L mutations. MET was also overexpressed as a positive control. Cells were seeded in 96-well plates and treated for 1 wk with increasing concentrations of Cetuximab (0–12.5 µg/mL) (left) or assayed using a clonogenic survival assay at 20 d at 1 or 10 µg/mL of Cetuximab (right). (C) Immunoblot of the effect of Cetuximab (10 µg/mL) at 6 h on pERK in MAP2K1 mutant CCK-81 cells (as in B).