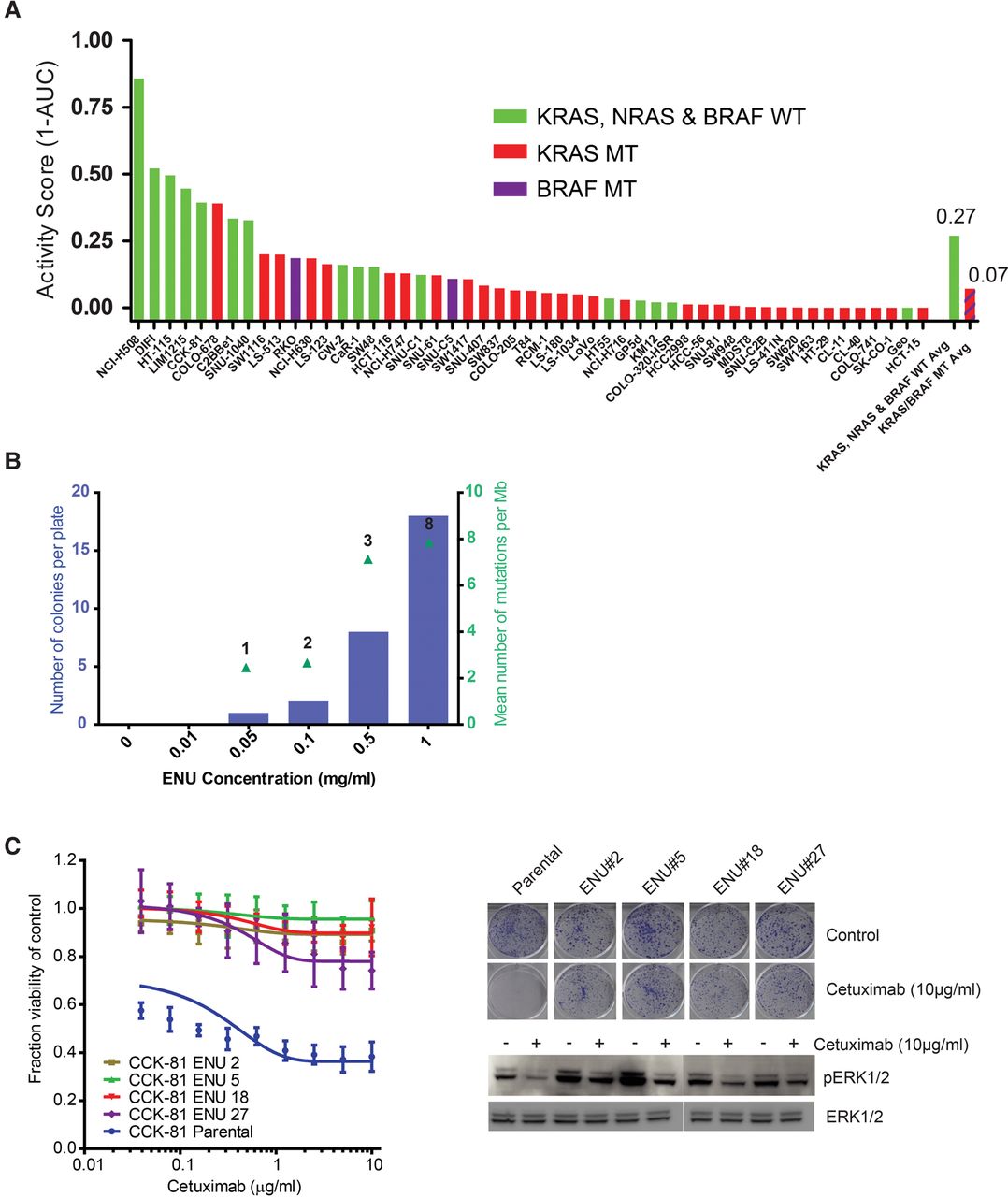
Genome-wide chemical mutagenesis screens to define pathways of drug resistance in cancer. (A) Cetuximab viability screen in colorectal cancer cell lines. Fifty-one colorectal cancer cell lines were screened with a concentration range of the EGFR monoclonal antibody Cetuximab and viability (as measured by the Activity Score, 1-AUC) was measured after 6 d. The KRAS, NRAS, and BRAF mutation status of each line is indicated. The mean Activity Score for those cell lines wild-type for all three genes (green bar) versus those with a canonical driver mutation in at least one gene (red bar) is indicated in the last two columns. (B) Dose-dependent effect of ENU on mutation burden and drug resistance. CCK-81 colorectal cancer cells were treated for 24 h with increasing concentrations of ENU (x-axis, 0.01–1 mg/mL) and then weekly with Cetuximab (10 µg/mL) for 8 wk to allow drug-resistant colonies to develop. The number of resistant colonies per plate were counted for each ENU concentration (blue bars) and submitted for whole-exome sequencing to calculate the mean number of mutations per Mb (green triangles). Numbers above each triangle indicate the number of clones sequenced at that concentration. (C) ENU mutagenesis generates stably drug-resistant clones. A subset of Cetuximab-resistant clones generated in the CCK-81 cell line following exposure to ENU and subsequent serial weekly Cetuximab treatment were picked from the plate and expanded separately out of drug for 4 wk. Twenty-one day clonogenic survival assays in four clones (along with the parental CCK-81 cell line) treated with Cetuximab 10 µg/mL confirmed that resistance to the EGFR monoclonal antibody had been maintained and was stable. A 6-d viability assay of cells treated with a concentration range of Cetuximab (right) demonstrates resistance of ENU clones at all concentrations. Immunoblot analysis of effect of Cetuximab treatment (10 µg/mL) for 6 h confirms persistence of MAPK signaling in ENU clones.