Abstract
EnteroBase is an integrated software environment that supports the identification of global population structures within several bacterial genera that include pathogens. Here, we provide an overview of how EnteroBase works, what it can do, and its future prospects. EnteroBase has currently assembled more than 300,000 genomes from Illumina short reads from Salmonella, Escherichia, Yersinia, Clostridioides, Helicobacter, Vibrio, and Moraxella and genotyped those assemblies by core genome multilocus sequence typing (cgMLST). Hierarchical clustering of cgMLST sequence types allows mapping a new bacterial strain to predefined population structures at multiple levels of resolution within a few hours after uploading its short reads. Case Study 1 illustrates this process for local transmissions of Salmonella enterica serovar Agama between neighboring social groups of badgers and humans. EnteroBase also supports single nucleotide polymorphism (SNP) calls from both genomic assemblies and after extraction from metagenomic sequences, as illustrated by Case Study 2 which summarizes the microevolution of Yersinia pestis over the last 5000 years of pandemic plague. EnteroBase can also provide a global overview of the genomic diversity within an entire genus, as illustrated by Case Study 3, which presents a novel, global overview of the population structure of all of the species, subspecies, and clades within Escherichia.
Epidemiological transmission chains of Salmonella, Escherichia, or Yersinia have been reconstructed with the help of single-nucleotide polymorphisms (SNPs) from hundreds or even thousands of core genomes (Zhou et al. 2013, 2014, 2018c; Langridge et al. 2015; Connor et al. 2016; Dallman et al. 2016; Wong et al. 2016; Ashton et al. 2017; Alikhan et al. 2018; Waldram et al. 2018; Worley et al. 2018; Johnson et al. 2019). However, the scale of these studies pales in comparison to the numbers of publicly available archives (e.g., NCBI Sequence Read Archive [SRA]) of short-read sequences of bacterial pathogens that have been deposited since the recent drop in price of high-throughput sequencing (https://www.genome.gov/sequencingcostsdata/). In October 2019, SRA contained genomic sequence reads from 430,417 Salmonella, Escherichia/Shigella, Clostridioides, Vibrio, and Yersinia. However, until very recently (Sanaa et al. 2019), relatively few draft genomic assemblies were publicly available, and even the current comparative genomic analyses in NCBI Pathogen Detection (https://www.ncbi.nlm.nih.gov/pathogens/) are restricted to relatively closely related genetic clusters. Since 2014, EnteroBase (https://enterobase.warwick.ac.uk) has attempted to address this gap for selected genera that include bacterial pathogens (Table 1). EnteroBase provides an integrated software platform (Fig. 1) that can be used by microbiologists with limited bioinformatic skills to upload short reads, assemble and genotype genomes, and immediately investigate their genomic relationships to all natural populations within those genera. These aspects have been illustrated by recent publications providing overviews of the population structures of Salmonella (Alikhan et al. 2018) and Clostridioides (Frentrup et al. 2019), a description of the GrapeTree GUI (Zhou et al. 2018a), and a reconstruction of the genomic history of the Salmonella enterica Para C Lineage (Zhou et al. 2018c). However, EnteroBase also provides multiple additional features, which have hitherto largely been promulgated by word of mouth. Here, we provide a high-level overview of the functionality of EnteroBase, followed by exemplary case studies of S. enterica serovar Agama, Yersinia pestis, and all of Escherichia.
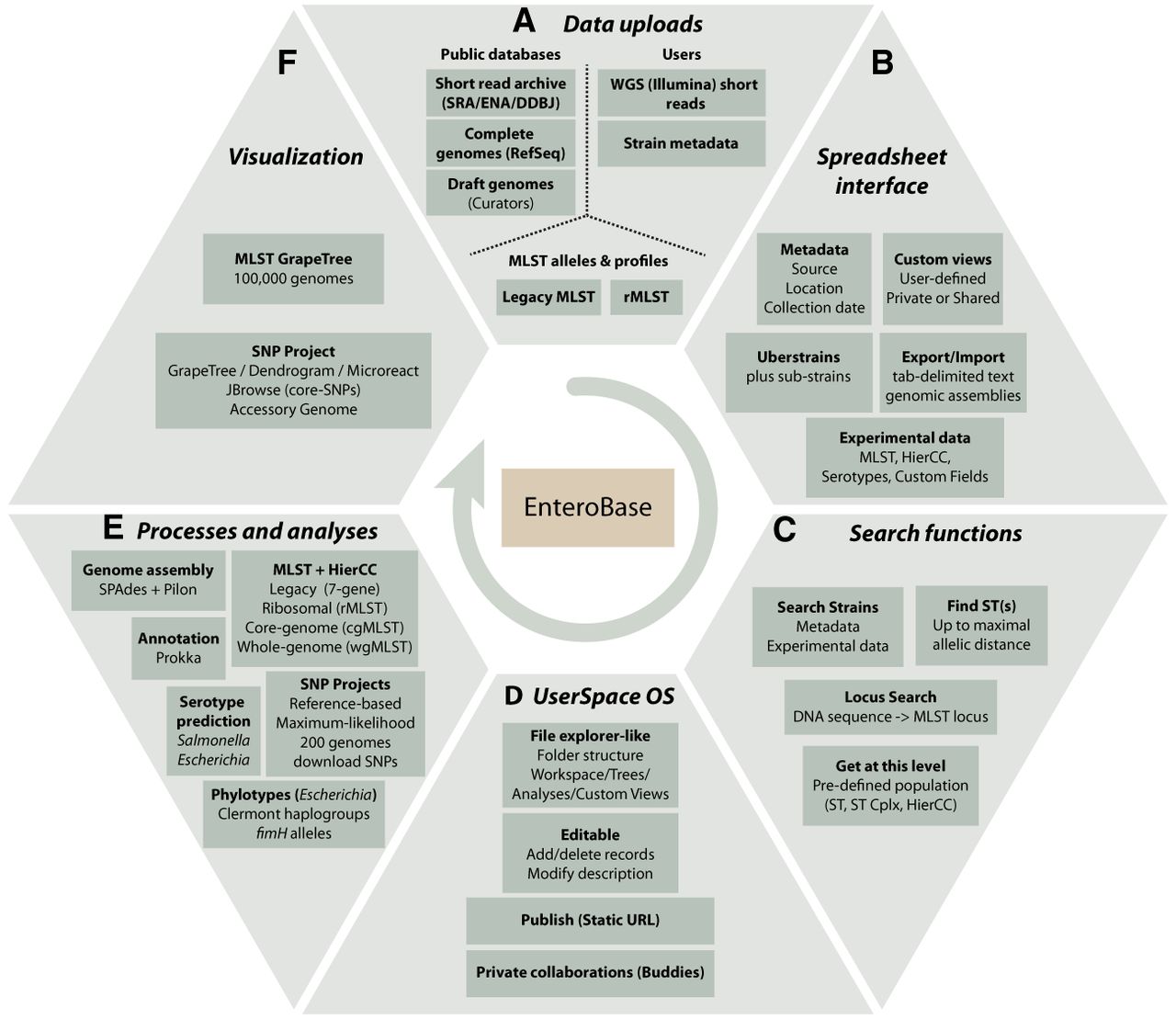
Overview of EnteroBase Features. (A) Data uploads. Data are imported from public databases, user uploads, and existing legacy MLST and rMLST databases at PubMLST (https://pubmlst.org/). (B) Spreadsheet Interface. The browser-based interface visualizes sets of strains (one Uberstrain plus any number of substrains) each containing metadata, and their associated experimental data and custom views. Post-release data can be exported (downloaded) as genome assemblies or tab-delimited text files containing metadata and experimental data. Metadata can be imported to entries for which the user has editing rights by uploading tab-delimited text files. (C) Search Strains supports flexible (AND/OR) combinations of metadata and experimental data for identifying entries to load into the spreadsheet. Find ST(s) retrieves STs that differ from a given ST by no more than a maximal number of differing alleles. Locus Search uses BLASTN (Altschul et al. 1990) and UBlastP in USEARCH (Edgar 2010) to identify the MLST locus designations corresponding to an input sequence. Get at this level: menu item after right clicking on experimental MLST ST or cluster numbers. (D) UserSpace OS. A file explorer–like interface for manipulations of workspaces, trees, SNP projects, and custom views. These objects are initially private to their creator but can be shared with buddies or rendered globally accessible. (E) Processes and analyses. EnteroBase uses EToKi and external programs as described in Supplemental Figure S1. (F) Visualization. MLST trees are visualized with the EnteroBase tools GrapeTree (Zhou et al. 2018a) and Dendrogram, which in turn can transfer data to external websites such as Microreact (Argimón et al. 2016).
Results
Overview of EnteroBase
The EnteroBase back end consists of multiple, cascading automated pipelines (Supplemental Fig. S1) that implement the multiple functions that it provides (Supplemental Fig. S2A). Many of these EnteroBase pipelines are also available within EToKi (EnteroBase ToolKit) (Supplemental Code), a publicly available repository (https://github.com/zheminzhou/EToKi) of useful modules (Supplemental Fig. S2B–E) that facilitate genomic assemblies (EToKi modules prepare and assemble), MLST (MLSType), calling nonrepetitive SNPs against a reference genome (EToKi modules align and phylo), or predicting serotypes of Escherichia coli from genome assemblies (EBEis).
EnteroBase performs daily scans of SRA via its Entrez APIs (Clark et al. 2016) for novel Illumina short-read sequences for each of the bacterial genera that it supports. It uploads the new reads and assembles them (EBAssembly [Supplemental Fig. S2B]) into annotated draft genomes, which are published if they pass quality control (Supplemental Table S1). EnteroBase fetches the metadata associated with the records and attempts to transcribe it automatically into EnteroBase metadata format (Supplemental Table S2; Supplemental Fig. S3). During the conversion, geographic metadata are translated into structured format using the Nominatim engine offered by OpenStreetMap (OpenStreetMap contributors 2017; Planet dump retrieved from https://planet.osm.org) and the host/source metadata are assigned to predefined categories (Supplemental Table S4). Until recently, metadata was parsed using a pretrained Native Bayesian classifier implemented in the Natural Language Toolkit (NLTK) for Python (Bird et al. 2009) with an estimated accuracy of 60%. Since November 2019, a new metaparser is being used, with an estimated accuracy of 93% (Supplemental Material), and all old data will soon be re-parsed. Registered users can upload their own Illumina short reads and metadata into EnteroBase; these are then processed with the same pipelines.
The annotated genomes are used to call alleles for multilocus sequence typing (MLST) (MLSType [Supplemental Fig. S2C]) and their sequence types (STs) are assigned to population groupings as described below. Salmonella serovars are predicted from the legacy MLST eBurstGroups (eBGs), which are strongly associated with individual serovars (Achtman et al. 2012), or by two external programs—SISTR1 (Yoshida et al. 2016; Robertson et al. 2018) and SeqSero2 (Zhang et al. 2019)—which evaluate genomic sequences. Escherichia serotypes are predicted from the genome assemblies by the EnteroBase module EBEis (Supplemental Fig. S2E). Clermont haplogroups are predicted for Escherichia by two external programs—ClermonTyping (Beghain et al. 2018) and EZClermont (Waters et al. 2018)—and fimH type by a third (FimTyper) (Roer et al. 2017). By default, all registered users have full access to strain metadata and the genome assemblies, predicted genotypes, and predicted phenotypes, but a delay in the release date of up to 12 mo can be imposed by users when uploading short-read sequences.
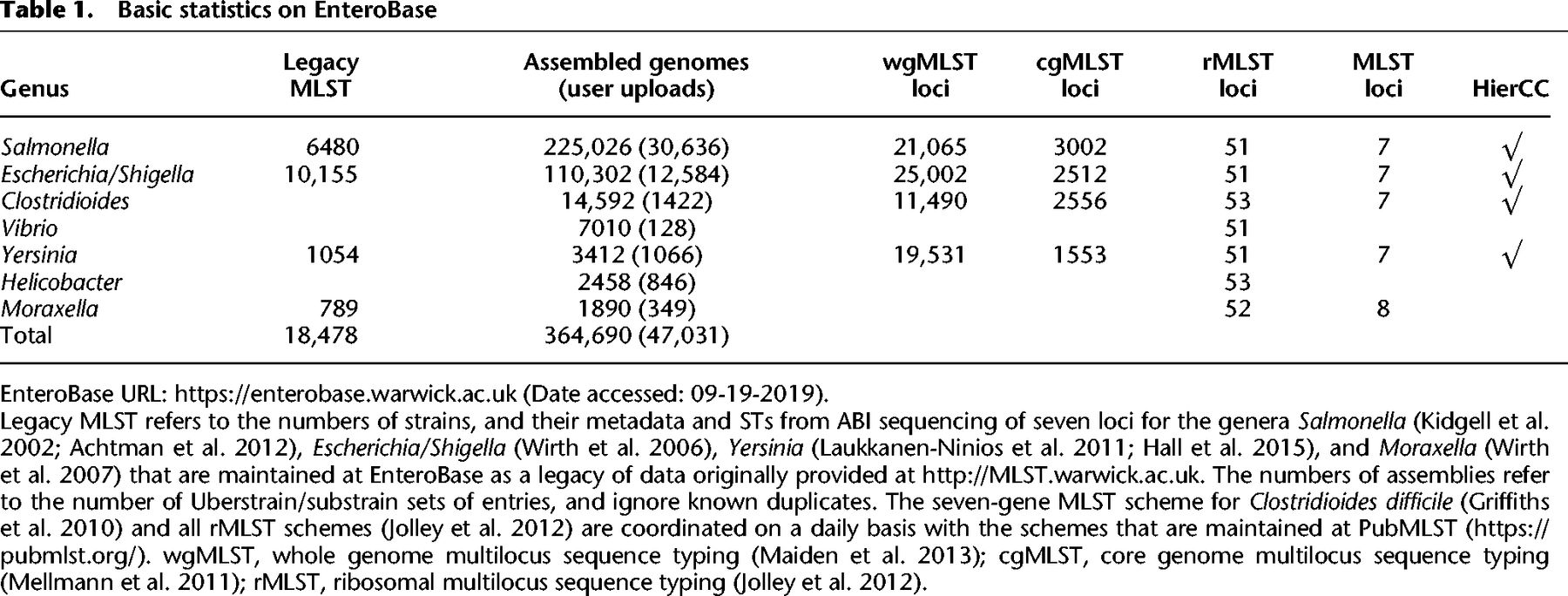
In September 2019, EnteroBase provided access to 364,690 genomes and their associated metadata and predictions (Table 1). To allow comparisons with historical data, EnteroBase also maintains additional legacy seven-gene MLST assignments (and metadata) that were obtained by classical Sanger sequencing from 18,478 strains.
Ownership, permanence, access, and privacy
EnteroBase users can upload new entries, consisting of paired-end Illumina short reads plus their metadata. Short reads are deleted after genome assembly, or after automated, brokered uploading of the reads and metadata to the European Nucleotide Archive (ENA) upon user request.
The search and graphical tools within EnteroBase can access all assembled genomes and their metadata, even if they are pre-release. However, ownership of uploaded data remains with the user and extends to all calculations performed by EnteroBase. Only owners and their buddies, administrators, or curators can edit the metadata; and only those individuals can download any data or calculations before their release date. To facilitate downloading of post-release data by the general community, downloads containing metadata and genotypes or genomic assemblies are automatically stripped of pre-release data for users who lack ownership privileges. Similarly, pre-release nodes within trees in the GrapeTree and Dendrogram graphical modules must be hidden before users without ownership privileges can download those trees.
In general, metadata that were imported from SRA are not editable, except by administrators and curators. However, the administrators can assign editing rights to users with claims to ownership or to those who possess special insights.
MLST population structures
Each unique sequence variant of a gene in an MLST scheme is assigned a unique numerical designation. Seven-gene MLST STs consist of seven integers for the alleles of seven housekeeping gene fragments (Maiden et al. 1998). rSTs consist of 51–53 integers for ribosomal protein gene alleles (Jolley et al. 2012). cgMLST STs consist of 1553–3002 integers for the number of genes in the soft core genome for that genus (Table 1), which were chosen as described elsewhere (Frentrup et al. 2019). However, STs are arbitrary constructs, and natural populations can each encompass multiple, related ST variants. Therefore, seven-gene STs are grouped into ST complexes in Escherichia/Shigella (Wirth et al. 2006) by an eBurst approach (Feil et al. 2004) and into their equivalent eBurst groups (eBGs) in Salmonella (Achtman et al. 2012). EnteroBase implements similar population groups (reBGs) for rMLST in Salmonella, which are largely consistent with eBGs or their subpopulations (Alikhan et al. 2018). The EnteroBase Nomenclature Server (Supplemental Fig. S1) calculates these population assignments automatically for each novel ST on the basis of single-linkage clustering chains with maximal pairwise differences of one allele for seven-gene MLST and two alleles for rMLST. To prevent overlaps between ST complexes, growing chains are terminated when they extend too closely to other existing populations (two allele difference in seven-gene MLST and five in rMLST).
cgMLST has introduced additional complexities over MLST and rMLST. Visual comparisons of cgSTs are tedious and rarely productive, because each consists of up to 3002 integers. Furthermore, almost all cgSTs contain some missing data because they are called from draft genomes consisting of multiple contigs. EnteroBase contains 100,000s of cgST numbers because almost every genome results in a unique cgST number, although many cgSTs differ from others only by missing data. EnteroBase supports working with so many cgSTs through Hierarchical Clustering (HierCC), a novel approach which supports analyses of population structures based on cgMLST at multiple levels of resolution. To identify the cutoff values in stepwise cgMLST allelic distances which would reliably resolve natural populations, we first calculated a matrix of pairwise allelic distances (excluding pairwise missing data) for all existing pairs of cgSTs, and one matrix for the HierCC cluster numbers at each level of allelic distance, that is, one matrix for HC0, HC1, HC2, …, HC3001. A genus-specific subset of the most reliable HierCC clusters is reported by EnteroBase.
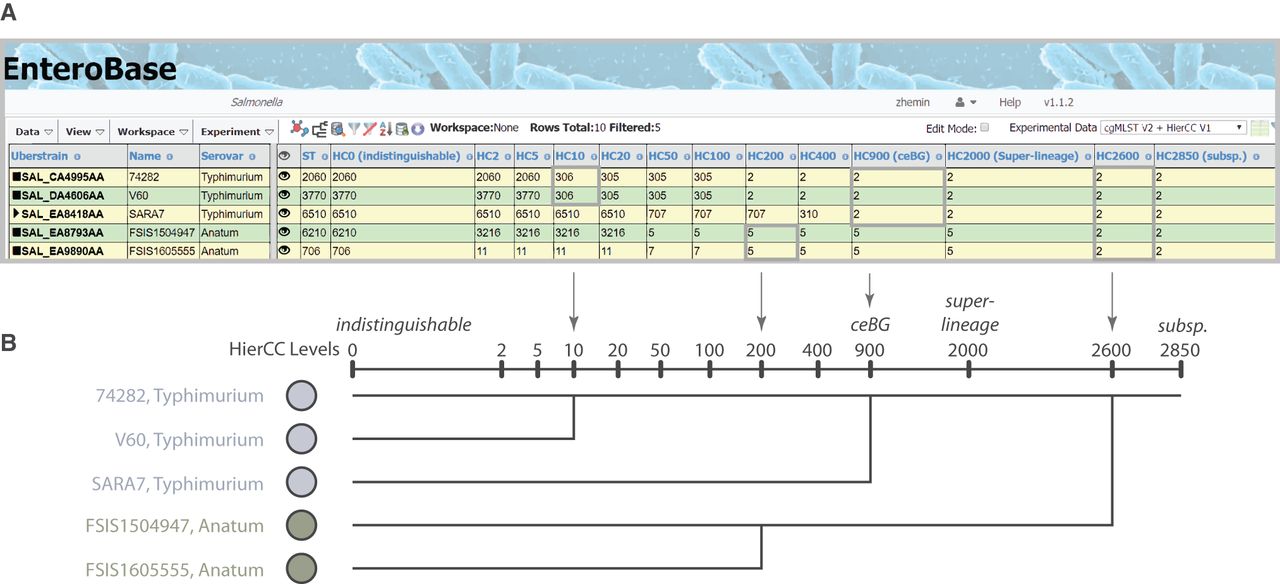
For Salmonella, 13 HierCC levels are reported, ranging from HC0 (indistinguishable except for missing data) to HC2850 (Fig. 2). Our experience with Salmonella indicates that HC2850 corresponds to subspecies, HC2000 to superlineages (Zhou et al. 2018c), and HC900 to cgMLST versions of eBGs. Long-term endemic persistence seems to be associated with HC100 or HC200; and epidemic outbreaks with HC2, HC5, or HC10. Eleven levels are reported for the other genera, ranging from HC0 up to HC2350 for Escherichia, HC2500 for Clostridioides, and HC1450 for Yersinia. Escherichia HC1100 corresponds to ST Complexes (see below) and the correspondences to population groupings in Clostridioides are described elsewhere (Frentrup et al. 2019). Further information on HierCC can be found in the EnteroBase documentation (https://enterobase.readthedocs.io/en/latest/features/clustering.html).
The hierarchical cgMLST clustering (HierCC) scheme in EnteroBase. (A) A screenshot of Salmonella cgMLST V2 plus HierCC V1 data for five randomly selected genomes. The numbers in the columns are the HierCC cluster numbers. Cluster numbers are the smallest cgMLST ST number in single-linkage clusters of pairs of STs that are joined by up to the specified maximum number of allelic differences. These maximum differences are indicated by the suffix of each HC column, starting with HC0 for 0 cgMLST allelic differences, other than missing data, through to HC2850 for 2850 allelic differences. The cluster assignments are greedy because individual nodes which are equidistant from multiple clusters are assigned to the cluster with the smallest cluster number. (B) Interpretation of HierCC numbers. The assignments of genomic cgMLST STs to HC levels can be used to assess their genomic relatedness. The top two genomes are both assigned to HC10_306, which indicates a very close relationship, and may represent a transmission chain. The top three genomes are all assigned to HC900_2, which corresponds to a legacy MLST eBG. HC2000 marks superlineages (Zhou et al. 2018c), and HC2850 marks subspecies. This figure illustrates these interpretations in the form of a cladogram drawn by hand.
Uberstrains and substrains
Most bacterial isolates/strains in EnteroBase are linked to one set of metadata and one set of genotyping data. However, EnteroBase includes strains for which legacy MLST data from classical Sanger sequencing exists in addition to MLST genotypes from genomic assemblies. Similarly, some users have uploaded the same reads to both EnteroBase and SRA, and both sets of data are present in EnteroBase. In other cases, genomes of the same strain have been sequenced by independent laboratories, or multiple laboratory variants have been sequenced that are essentially indistinguishable (e.g., S. enterica LT2 or E. coli K-12).
EnteroBase deals with such duplicates by implementing the concept of an Uberstrain, which can be a parent to one or more identical substrains. Substrains remain invisible unless they are specified in the search dialog (Supplemental Fig. S4), in which case they are shown with a triangle in the Uberstrain column (Fig. 3A). Examples of the usage of this approach can be found in Supplemental Material.
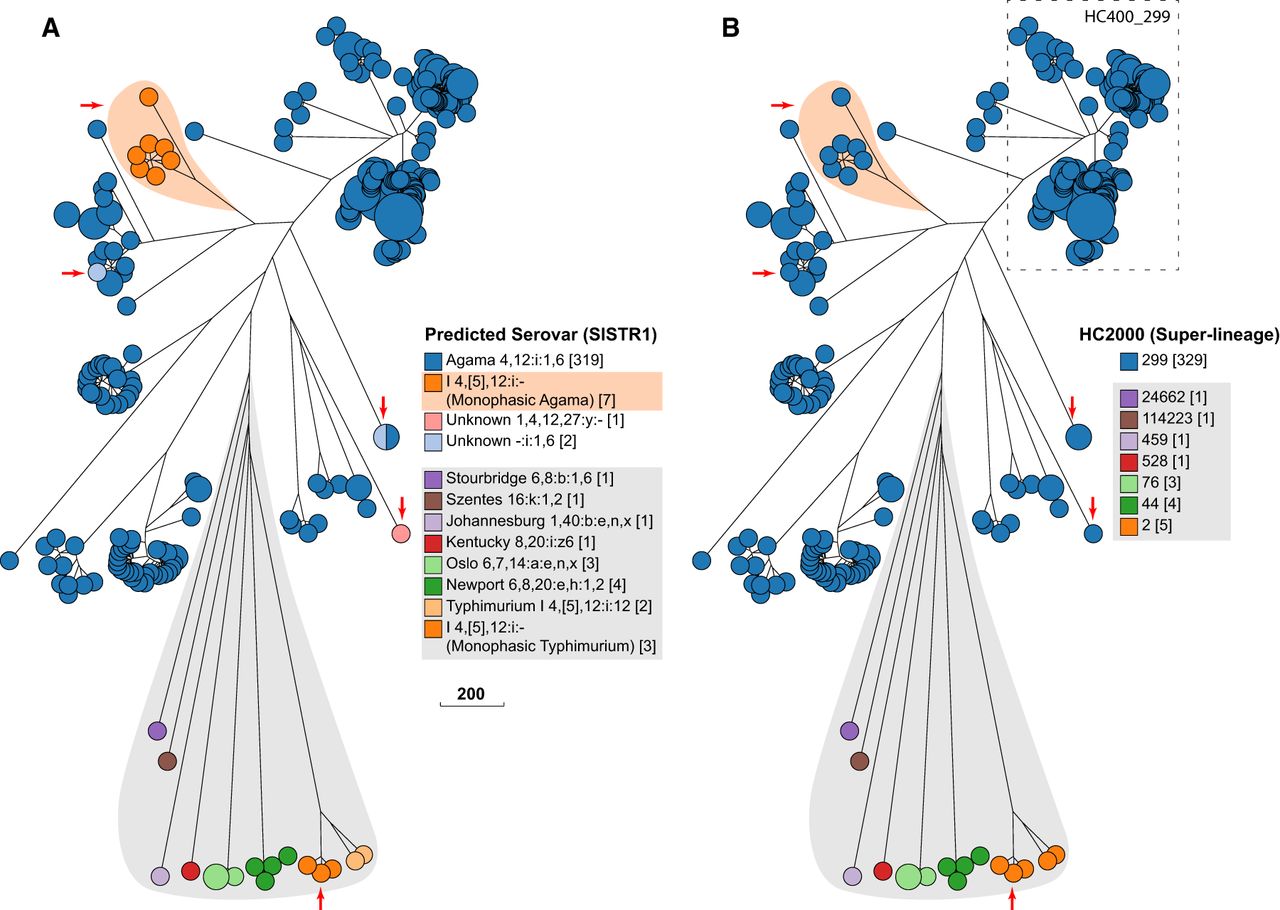
Serovar versus HierCC clustering in serovar Agama. GrapeTree (Zhou et al. 2018a) depiction of a RapidNJ tree (Simonsen et al. 2011) of cgMLST allelic distances between genomic entries whose metadata Serovar field contained Agama or SISTR1 (Robertson et al. 2018) Serovar predictions contained Agama. (A) Color coding by Predicted Serovar (SISTR1). Arrows indicate isolates whose serovar was not predicted. Orange shading emphasizes 1,4,[5],12:i:- isolates that were monophasic Agama. Gray shading indicates isolates with incorrect Serovar metadata, including 1,4,[5],12:i:- isolates that were monophasic Typhimurium (arrow). (B) Color coding by HC2000 cluster. All Agama entries are HC2000_299, as were the genetically related entries marked with arrows or emphasized by orange shading. Entries from other serovars (gray shading) were in other diverse HC2000 clusters. The dashed box indicates a subset of Agama strains within HC400_299, including all isolates from badgers, which were chosen for deeper analyses in Figure 4. (Scale bar) Number of cgMLST allelic differences.
Examples of the utility of EnteroBase
Often the utility of a tool first becomes clear through examples of its use. Here, we present three case studies that exemplify different aspects of EnteroBase. Case Study 1 shows how geographically separated laboratories can collaborate in private on an EnteroBase project until its completion, upon which EnteroBase publishes the results. This example focuses on geographical microvariation and transmission chains between various host species of a rare serovar of S. enterica. Case Study 2 shows how to combine modern genomes of Yersinia pestis with partially reconstructed genomes from ancient skeletons of plague victims. It also shows how EToKi can extract SNPs from metagenomic sequence reads. Case Study 3 provides a detailed overview of the genomic diversity of the genus Escherichia and defines the EcoRPlus set of representative genomes.
Case Study 1: a group collaboration on S. enterica serovar Agama
S. enterica subspecies enterica encompasses more than 1586 defined serovars (Guibourdenche et al. 2010; Issenhuth-Jeanjean et al. 2014). These differ in the antigenic formulas of their lipopolysaccharide (O antigen) and/or two alternative flagellar antigens (H1, H2), which are abbreviated as O:H1:H2. Some serovars are commonly isolated from infections and the environment and have been extensively studied. Others are rare, poorly understood, and often polyphyletic (Achtman et al. 2012), including Salmonella that colonize badgers (Wray et al. 1977; Wilson et al. 2003).
In late 2018, serovar Agama (antigenic formula: 4,12:i:1,6) was specified in the Serovar metadata field for only 134/156,347 (0.09%) genome assemblies in EnteroBase, and all 134 isolates were from humans. We were therefore interested to learn that the University of Liverpool possessed serovar Agama isolates that had been isolated in 2006–2007 from European badgers (Meles meles) in Woodchester Park, Gloucestershire, England. We sequenced the genomes of 72 such isolates and uploaded the short reads and strain metadata into EnteroBase. This data was used to analyze the population structure of a rare serovar within a single host species over a limited geographical area and to compare Agama genomes from multiple hosts and geographical sources.
Search strains
The browser interface to EnteroBase is implemented as a spreadsheet-like window called a “Workspace” that can page through thousands of entries, showing metadata at the left and experimental data at the right (https://enterobase.readthedocs.io/en/latest/features/using-workspaces.html). However, visual scanning of so many entries is inefficient. EnteroBase therefore offers powerful search functions (https://enterobase.readthedocs.io/en/latest/enterobase-tutorials/search-agama.html) for identifying isolates that share common phenotypes (metadata) and/or genotypes (experimental data).
EnteroBase also predicts serovars from assembled Salmonella genomes and from MLST data. However, the software predictions are not failproof, and many entries lack metadata information or the metadata is erroneous. We therefore used the Search Strains dialog box to find entries containing “Agama” in the metadata Serovar field or by the Serovar predictions from SISTR1 (https://enterobase.readthedocs.io/en/latest/enterobase-tutorials/search-agama.html). Phylogenetic analyses of the cgMLST data from those entries indicated that Agama consisted of multiple microclusters.
International participation in a collaborative network
Almost all Agama isolates in EnteroBase were from England, which represents a highly skewed geographical sampling bias that might lead to phylogenetic distortions. We therefore formed the Agama Study Group, consisting of colleagues at national microbiological reference laboratories in England, Scotland, Ireland, France, Germany, and Austria. The participants were declared as “buddies” within EnteroBase (https://enterobase.readthedocs.io/en/latest/features/buddies.html) with explicit rights to access the Workspaces and phylogenetic trees in the Workspace\Load\Shared\Zhemin\Agama folder. After completion of this manuscript, that folder was made publicly available.
We facilitated the analysis of the Agama data by creating a new user-defined Custom View (https://enterobase.readthedocs.io/en/latest/features/user-defined-content.html), which can aggregate various sources of experimental data as well as user-defined fields. The Custom View was saved in the Agama folder, and thereby shared with the study group. It too was initially private but became public together with the other workspaces and trees when the folder was made public.
Members of the Agama Study Group were requested to sequence genomes from all Agama strains in their collections, and to upload those short reads to EnteroBase, or to send their DNAs to University of Warwick for sequencing and uploading. The new entries were added to the “All Agama Strains” workspace. The final set of 345 isolates had been isolated in Europe, Africa, and Australia, with collection years ranging from 1956 to 2018 (Supplemental Table S3).
Global population structure of Agama
We created a neighbor-joining GrapeTree (Zhou et al. 2018a) of cgMLST allelic differences to reveal the genetic relationships within serovar Agama. Color coding the nodes of the tree by SISTR1 serovar predictions confirmed that most isolates were Agama (Fig. 3A). However, one microcluster (shaded in light orange) consisted of seven monophasic Agama isolates with a defective or partial fljB (H2) CDS, which prevented a serovar prediction. SISTR1 also could not predict the O antigens of three other related isolates (arrows in Fig. 3). Sixteen other isolates on long branches were assigned to other serovars by SISTR1 (Fig. 3A, gray shading). Comparable results were obtained with SeqSero2 or eBG serovar associations, and these 16 isolates represent erroneous Serovar assignments within the metadata. Three of these erroneous Agama had the same predicted antigenic formula (1,4,[5],12:i:-) as the monophasic Agama isolates (orange shading), but these represent monophasic Typhimurium.
In contrast to serovar, coloring the tree nodes by HC2000 clusters (Fig. 3B) immediately revealed that all genomes that were called Agama by SISTR1 belonged to HC2000 cluster number 299 (HC2000_299), and all HC2000_299 were genetically related and clustered together in the tree (Fig. 3B). In contrast, the 16 other isolates on long branches (gray shading) belonged to other HC2000 clusters.
These results show that Agama belongs to one superlineage, HC2000_299, which has been isolated globally from humans, badgers, companion animals, and the environment since at least 1956. The genetic relationships would not have been obvious with lower resolution MLST: Some Agama isolates belong to eBG167, others to eBG336, and 13 Agama MLST STs do not belong to any eBG.
Transmission patterns at different levels of HierCC resolution
All isolates from badgers were in HierCC cluster HC400_299 (Fig. 3B, dashed box), which also included other isolates from humans and other animals. HC400_299 was investigated by maximum-likelihood trees of core, nonrepetitive SNPs called against a reference draft genome with the help of the EnteroBase Dendrogram GUI. One tree (Fig. 4A) encompassed 149 isolates from the British Isles which were in EnteroBase before establishing the Agama Study Group. A second tree (Fig. 4B) contained the final data set of 213 genomes, including isolates from additional badgers and multiple countries. A comparison of the two trees is highly instructive on the effects of sample bias.
Effects of sample bias on inferred transmission chains within HC400_299 Agama isolates. (A, left) Map of hosts in the British Isles of 149 Agama isolates in EnteroBase in August, 2018. (Right) Maximum-likelihood radial phylogeny (http://enterobase.warwick.ac.uk/a/21773/d) based on RAxML (Stamatakis 2014) of 8791 nonrepetitive core SNPs as calculated by EnteroBase Dendrogram against reference genome 283179. Color coding is according to a user-defined field (Location & Source). HC100 cluster designations for three microclades are indicated. HC100_2433 contained all Agama from badgers. (B, right) Summary of hosts and countries from which 64 additional Agama isolates had been sequenced by March 2019. (Left) Maximum-likelihood radial dendrogram (http://enterobase.warwick.ac.uk/a/23882/d) based on 9701 SNPs from 213 isolates. Multiple isolates of Agama in HC100_2433 were now from humans and food in France and Austria. HC100_299 and HC100_67355 now contained multiple isolates from badgers, livestock, companion animals, and mussels, demonstrating that the prior strong association of Agama with humans and badgers in A reflected sample bias. Stars indicate multiple MRCAs of Agama in English badgers, whereas the pink arrow indicates a potential transmission from badgers to a human in Bath/North East Somerset, which is close to Woodchester Park. The green arrow indicates a potential food-borne transmission chain consisting of four closely related Agama isolates in HC5_140035 from Austria (chives × 2; human blood culture × 1) and France (human × 1) that were isolated in 2018. The geographical locations of the badger isolates are shown in Supplemental Figure S5.
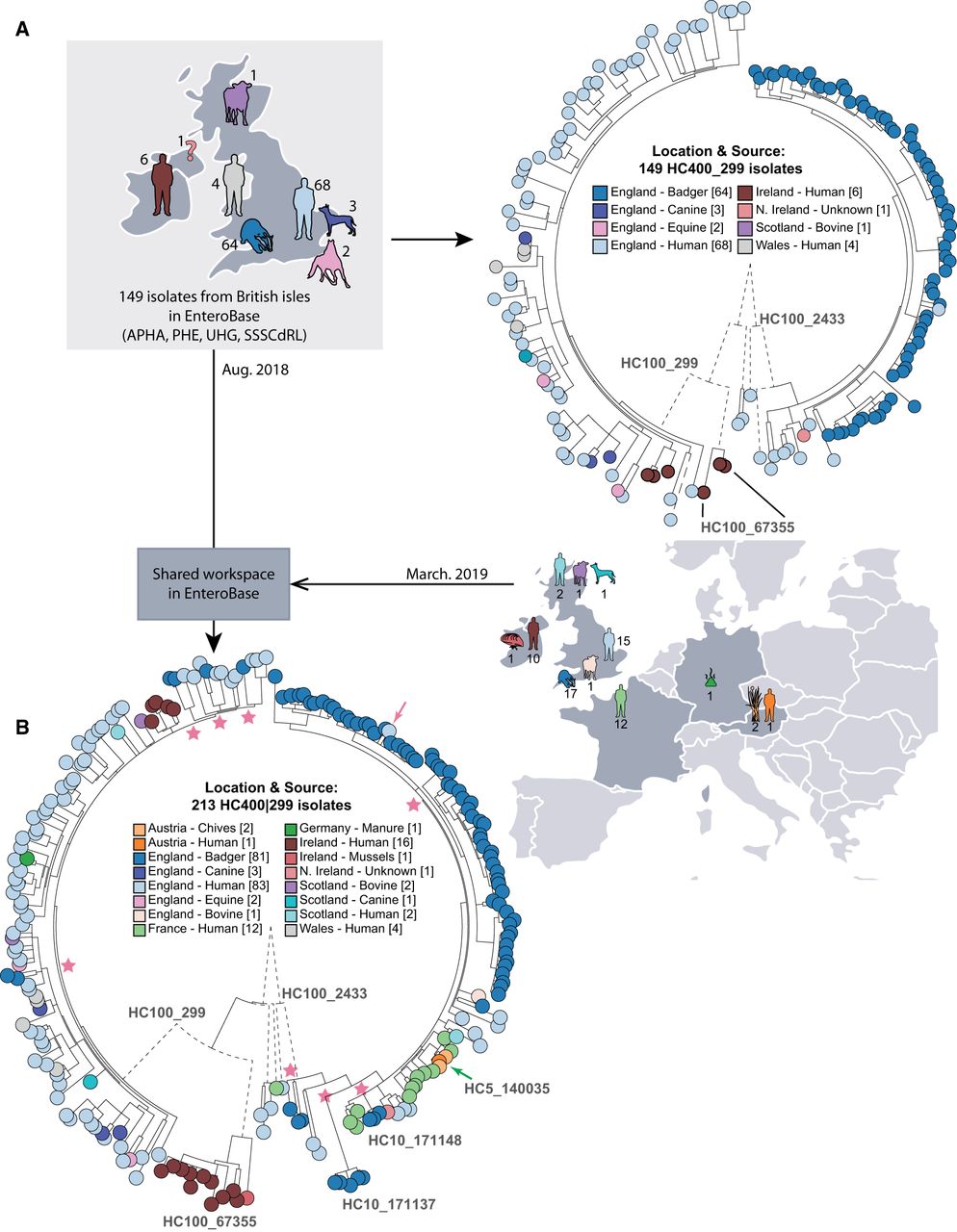
Almost all of the initial HC400_299 genomes fell into three clades designated HC100_299, HC100_2433, and HC100_67355. All badger isolates were from Woodchester Park (2006–2007) within the context of a long-term live capture–mark–recapture study (McDonald et al. 2018). The Agama isolates from those badgers formed a monophyletic subclade within HC100_2433, whose basal nodes represented human isolates. This branch topology suggested that a single recent common ancestor of all badger isolates had been transmitted from humans or their waste products.
The badgers in Woodchester Park occupy adjacent social group territories, which each contain several setts (burrows). HC100_2433 contains multiple HC10 clusters of Agama from badgers (Supplemental Fig. S5A). To investigate whether these microclusters might mark transmission chains between setts and social groups, a Newick subtree of HC100_2433 plus geographical coordinates was transmitted from GrapeTree to Microreact (Argimón et al. 2016), an external program which is specialized in depicting geographical associations. Badgers occasionally move between neighboring social groups (Rogers et al. 1998). Transmissions associated with such moves are supported by the observation that five distinct HC10 clusters each contained isolates from two social groups in close proximity (Supplemental Fig. S5B).
Long-term dispersals and interhost transmissions
The 63 additional HC400_299 Agama genomes that were sequenced by the Agama Study Group provided important insights on the dissemination of Agama over a longer time frame and showed the problems that can result from sample bias. Seventeen Agama strains had been isolated from English badgers at multiple locations in southwest England between 1998 and 2016 (Supplemental Fig. S5B) and stored at APHA. Eleven of them were in HC100_2433. However, rather than being interspersed among the initial genomes from badgers, they defined novel microclusters, including HC10_171137 and HC10_171148, which were the most basal clades in HC100_2433 (Fig. 4B). The other six badger isolates were from additional geographical sources and interspersed among human isolates in HC100_299 (Fig. 4B), which had previously not included any badger isolates (Supplemental Fig. S5F). These results show that the diversity of Agama from English badgers is comparable to their diversity within English humans, and that it would be difficult to reliably infer the original host of these clades or the directionality of interhost transmissions. Further observations on microepidemiology of Agama transmissions between hosts and countries are presented in Supplemental Material.
Case Study 2: combining modern Y. pestis genomes with ancient metagenomes
EnteroBase automatically scours sequence read archives for Illumina short reads from cultivated isolates, assembles their genomes, and publishes draft assemblies that pass quality control. In October 2019, EnteroBase had assembled more than 1300 genomes of Y. pestis, including genomes that had already been assigned to population groups (Cui et al. 2013), other recently sequenced genomes from central Asia (Eroshenko et al. 2017; Kutyrev et al. 2018), and numerous unpublished genomes from Madagascar and Brazil. EnteroBase does not upload assembled genomes, for which adequate, automated quality control measures would be difficult to implement. However, EnteroBase administrators can upload such genomes after ad hoc assessment of sequence quality, and EnteroBase contains standard complete genomes such as CO92 (Parkhill et al. 2001) and other genomes used to derive the Y. pestis phylogeny (Morelli et al. 2010).
EnteroBase also does not automatically assemble genomes from metagenomes containing mixed reads from multiple taxa, but similar to complete genomes, administrators can upload reconstructed ancient genomes derived from SNP calls against a reference genome.
Ancient Y. pestis
The number of publications describing ancient Y. pestis genomes has increased over the last few years as ancient plague has been progressively deciphered (Bos et al. 2011, 2016; Wagner et al. 2014; Rasmussen et al. 2015; Feldman et al. 2016; Spyrou et al. 2016, 2018; Margaryan et al. 2018; Namouchi et al. 2018; Keller et al. 2019; Spyrou et al. 2019). The metagenomic short reads used to reconstruct these genomes are routinely deposited in the public domain, but the reconstructed ancient genomes are not. This practice has made it difficult for non-bioinformaticians to evaluate the relationships between ancient and modern genomes from Y. pestis. However, EnteroBase now provides a solution to this problem.
The EnteroBase EToKi calculation package (Supplemental Code) can reconstruct an ancient genome assembly by unmasking individual nucleotides in a fully masked reference genome based on reliable SNP calls from metagenomic data (Supplemental Fig. S6). We ran EToKi on 56 published ancient metagenomes containing Y. pestis, and the resulting assemblies and metadata were uploaded to EnteroBase. EnteroBase users can now include those ancient genomes together with other reconstructed genomes and modern genomic assemblies in a workspace of their choice and use the EnteroBase SNP dendrogram module to calculate and visualize a maximum-likelihood tree (of up to a current maximum of 200 genomes).
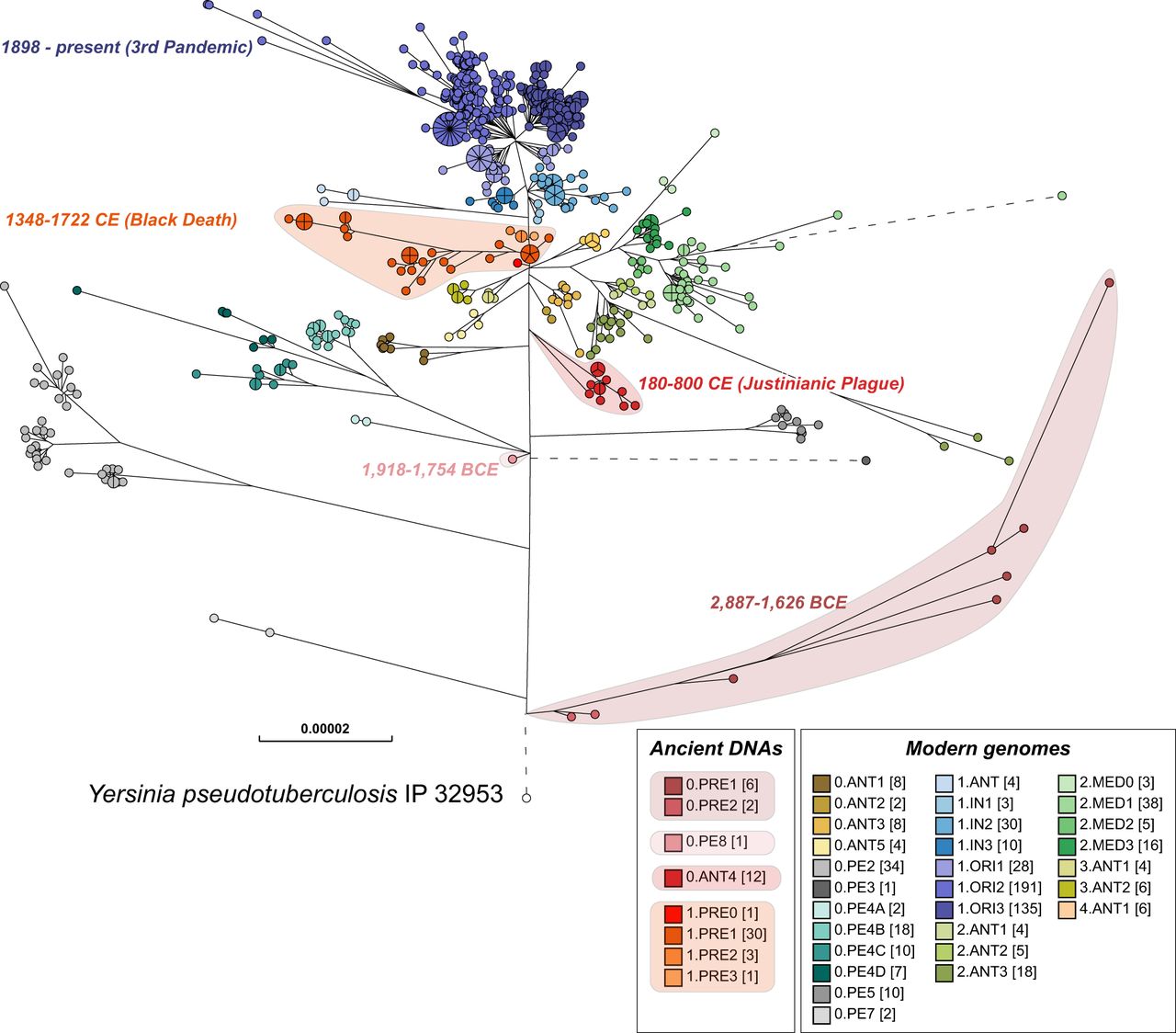
Figure 5 presents a detailed overview of the genomic relationships of all known Y. pestis populations from pandemic plague over the last 5500 years, including hundreds of unpublished modern genomes. This tree was manually annotated using a User-defined Field and Custom View with population designations for reconstructed ancient genomes that are consistent with the literature on modern isolates. We also assigned consistent population designations to additional modern genomes from central Asia and elsewhere. An interactive version of this tree and all related metadata in EnteroBase is publicly available (http://enterobase.warwick.ac.uk/a/21977/g), thus enabling its detailed interrogation by a broad audience from multiple disciplines (Green 2018) and providing a common language for scientific discourse.
Maximum-likelihood tree of modern and ancient genomes of Y. pestis. EnteroBase contained 1368 ancient and modern Y. pestis genomes in October 2019, of which several hundred genomes that had been isolated in Madagascar and Brazil over short time periods showed very low levels of genomic diversity. To reduce this sample bias, the data set used for analysis included only one random representative from each HC0 group from those two countries, leaving a total of 622 modern Y. pestis genomes. Fifty-six ancient genomes of Y. pestis from existing publications were assembled with EToKi (Methods), resulting in a total of 678 Y. pestis genomes plus Yersinia pseudotuberculosis IP32953 as an outgroup (http://enterobase.warwick.ac.uk/a/21975). The EnteroBase pipelines (Supplemental Fig. S2D) were used to create a SNP project in which all genomes were aligned against CO92 (2001) using LASTAL. The SNP project identified 23,134 nonrepetitive SNPs plus 7534 short inserts/deletions over 3.8 Mbps of core genomic sites which had been called in ≥95% of the genomes. In this figure, nodes are color coded by population designations for Y. pestis according to published sources (Morelli et al. 2010; Cui et al. 2013; Achtman 2016), except for 0.PE8 which was assigned to a genome from 1918 to 1754 BCE (Spyrou et al. 2018). The designation 0.ANT4 was applied by Achtman (2016) to Y. pestis from the Justinianic plague described by Wagner et al. (2014), and that designation was also used for a genome associated with the Justinianic plague (DA101) that was later described by Damgaard et al. (2018) as 0.PE5.
Case Study 3: Thinking big. An overview of the core genomic diversity of Escherichia/Shigella
Escherichia coli has long been one of the primary workhorses of molecular biology. Most studies of Escherichia have concentrated on a few well-characterized strains of E. coli, but the genus Escherichia includes other species: E. fergusonii, E. albertii, E. marmotae (Liu et al. 2015), and E. ruysiae (van der Putten et al. 2019). E. coli itself includes the genus Shigella (Pupo et al. 2000), which was assigned a distinctive genus name because it causes dysentery. Initial analyses of the phylogenetic structure of E. coli identified multiple deep branches, called haplogroups (Selander et al. 1987), and defined the EcoR collection (Ochman and Selander 1984), a classical group of 72 bacterial strains that represented the genetic diversity found with multilocus enzyme electrophoresis. The later isolation of environmental isolates from lakes revealed the existence of “cryptic clades” I–VI which were distinct from the main E. coli haplogroups and the other Escherichia species (Walk et al. 2009; Luo et al. 2011). Currently, bacterial isolates are routinely assigned to haplogroups or clades by PCR tests for the presence of variably present genes from the accessory genome (Clermont et al. 2013) or by programs that identify the presence of those genes in genomic sequences (Beghain et al. 2018; Waters et al. 2018).
Legacy MLST is an alternative scheme for subdividing Escherichia, which includes the assignment of STs to ST Complexes (Wirth et al. 2006). Several ST Complexes are common causes of invasive disease in humans and animals, such as ST131 (Stoesser et al. 2016; Liu et al. 2018), ST95 Complex (Wirth et al. 2006; Gordon et al. 2017), and ST11 Complex (O157:H7) (Eppinger et al. 2011a,b; Newell and La Ragione 2018). The large number of Escherichia genomes in EnteroBase (Table 1) offered the opportunity to reinvestigate the population structure of Escherichia on the basis of the greater resolution provided by cgMLST and within the context of a much larger and more comprehensive sample. In 2018, EnteroBase contained 52,876 genomes. To render this sample amenable to calculating a maximum-likelihood (ML) tree of core SNPs, we selected a representative sample consisting of one genome from each of the 9479 Escherichia rSTs. In homage to the EcoR collection, we designate this as the EcoRPlus Collection.
Core genome genetic diversity within Escherichia
Homologous recombination is widespread within E. coli (Wirth et al. 2006). We therefore anticipated that a phylogenetic tree of core genomic differences in EcoRPlus would be “fuzzy,” and that ST Complexes and other genetic populations would be only poorly delineated. Instead, considerable core genome population structure is visually apparent in a RapidNJ tree based on pairwise differences at cgMLST alleles between the EcoRPlus genomes (Fig. 6). The most predominant, discrete sets of node clusters were also largely uniform according to cgMLST HC1100 hierarchical clustering. Furthermore, assignments to HC1100 clustering were also largely congruent with ST Complexes based on legacy seven-gene MLST (Supplemental Fig. S7). With occasional exceptions (arrows), the tree topology was also consistent with Clermont typing (Supplemental Fig. S8; Supplemental Material).
Neighbor-joining (RapidNJ) tree of core genome allelic distances in the EcoRPlus Collection of 9479 genomes. EcoRPlus includes the draft genome with the greatest N50 value from each of the 9479 rSTs among 52,876 genomes of Escherichia within EnteroBase (August 2018) (http://enterobase.warwick.ac.uk/a/15931). The nodes in this tree are color coded by HC1100 clusters, as indicated in the key at the bottom left. Common HC1100 clusters (plus the corresponding ST Complexes) are indicated at the circumference of the tree. These are largely congruent, except that HC1100_13 corresponds to ST10 Complex plus ST168 Complex, and other discrepancies exist among the smaller, unlabeled populations. See Supplemental Figures S7, S8, respectively, for color coding by ST Complex and Clermont typing. An interactive version in which the nodes can be freely color coded by all available metadata is available at http://enterobase.warwick.ac.uk/a/15981. A maximum-likelihood tree based on SNP differences can be found in Supplemental Figure S9.
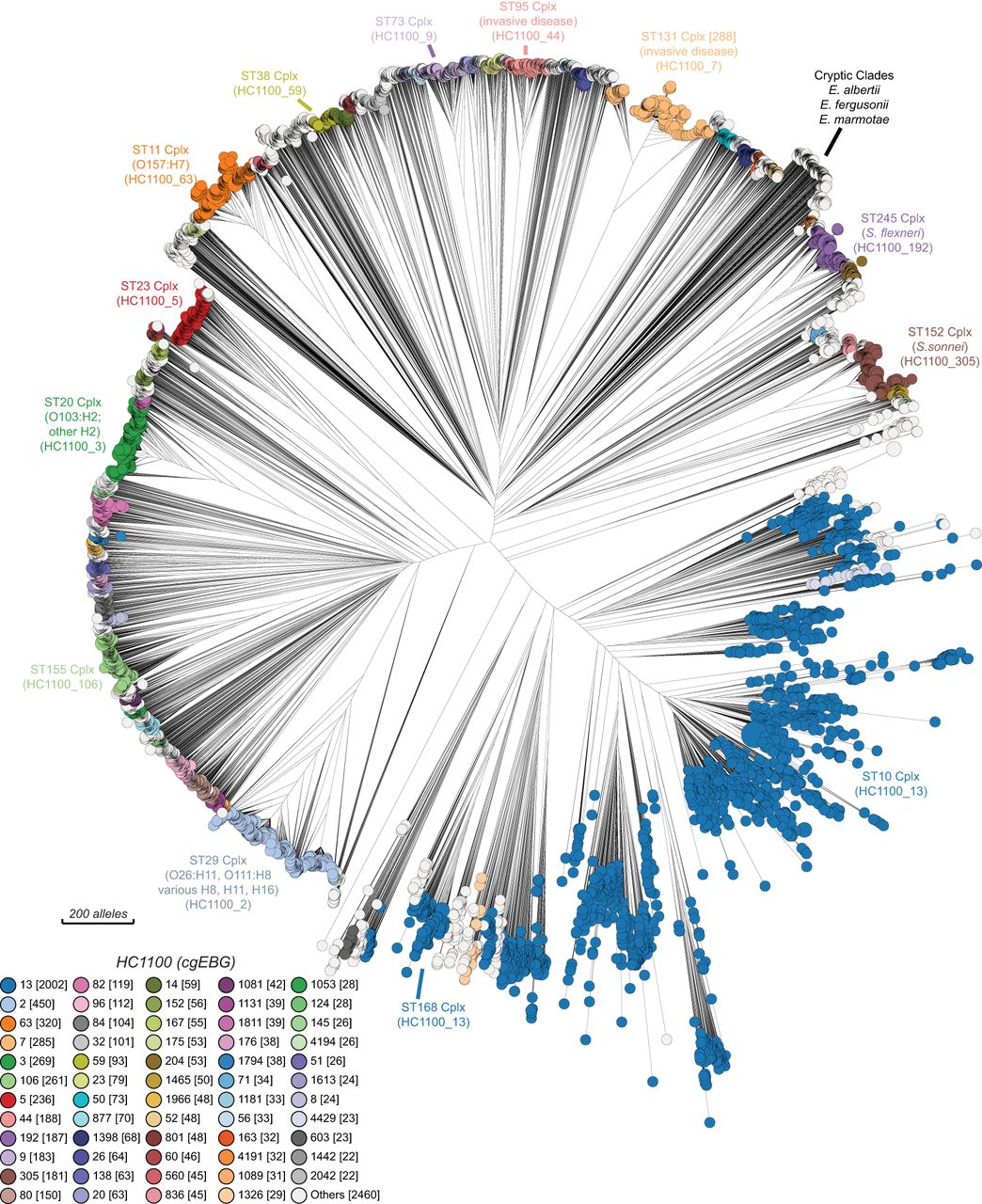
Figure 6 may represent the first detailed overview of the genetic diversity of the core genome of Escherichia. Real-time examination of its features (http://enterobase.warwick.ac.uk/a/15981) is feasible because the GrapeTree algorithm can handle large numbers of cgSTs (Zhou et al. 2018a). Nodes can be readily colored by metadata or experimental data (Supplemental Figs. S7, S8), and GrapeTree also readily supports analyses of subtrees in greater detail. However, although cgMLST allelic distances are reliable indicators of population structures, SNPs are preferable for examining genetic distances. We therefore calculated a ML tree of the 1,230,995 core SNPs within all 9479 genomes (Supplemental Fig. S9). This tree confirmed the clustering of the members of HC1100 groups within E. coli, and also showed that the other Escherichia species and cryptic clades II to VIII formed distinct long branches of comparable lengths (Supplemental Fig. S9 inset).
Discussion
EnteroBase was originally developed as a genome-based successor to the legacy MLST websites for Escherichia (Wirth et al. 2006), Salmonella (Achtman et al. 2012), Yersinia pseudotuberculosis (Laukkanen-Ninios et al. 2011), and Moraxella catarrhalis (Wirth et al. 2007). Its underlying infrastructure is sufficiently generic that EnteroBase was readily extended to Clostridioides, Helicobacter, and Vibrio, and could in principle be extended to other taxa.
EnteroBase was intended to provide a uniform and reliable pipeline that can assemble consistent draft genomes from the numerous short-read sequences in public databases (Achtman and Zhou 2014) and to link those assemblies with metadata and genotype predictions. It was designed to provide access to an unprecedentedly large global set of draft genomes to users at both extremes of the spectrum of informatics skills. A further goal was to provide analytical tools, such as GrapeTree (Zhou et al. 2018a), that could adequately deal with cgMLST from more than 100,000 genomes, and Dendrogram, which generates phylograms from nonrepetitive core SNPs called against a reference genome. Still another important goal was to support private analyses by groups of colleagues, with the option of subsequently making those analyses publicly available. Case Study 1 illustrates how EnteroBase can be used for all of these tasks, and more.
EnteroBase has expanded beyond its original goals and is morphing in novel directions. It has implemented HierCC for cgMLST, which supports the automated recognition of population structures at multiple levels of resolution (Case Study 1), and may help with the annotation of clusters within phylogenetic trees (Case Study 2; see below). EnteroBase has also been extended to support analyses of metagenomic data from ancient genomes (Zhou et al. 2018c; Achtman and Zhou 2019) by implementing a subset of the functionality of SPARSE (Zhou et al. 2018b) within the stand-alone EToKi package. Case Study 2 illustrates this capability for Y. pestis. Additional EnteroBase databases are under development for ancient and modern genomes of S. enterica and biofilms within dental calculus. EnteroBase has also shown its capacities for providing overviews of the core genome diversity of entire genera, with currently extant examples consisting of Salmonella (Alikhan et al. 2018) and Escherichia (Case Study 3).
EnteroBase is already being used by the community to identify genetically related groups of isolates (Diemert and Yan 2019; Haley et al. 2019; Johnson et al. 2019; Miller et al. 2019; Numberger et al. 2019), and HierCC has been used to mark international outbreaks of S. enterica serovar Poona (Jones et al. 2019b) and E. coli O26 (Jones et al. 2019a). Case Study 1 illustrates how to explore HierCC genomic relationships at multiple levels, ranging from HC2000 (superlineages) for intercontinental dispersion down to HC5-10 for detecting local transmission chains.
Case Study 1 confirms that although S. enterica serovar Agama is rare, it has been isolated from multiple hosts and countries and is clearly not harmless for humans. The results also document that an enormous sample bias exists in current genomic databases because they largely represent isolates that are relevant to human disease from a limited number of geographic locations.
Case Study 1 may also become a paradigm for identifying long-distance chains of transmission between humans or between humans and their companion or domesticated animals: Four Agama isolates in the HC5_140035 cluster from France (human) and Austria (frozen chives and a human blood culture) differed by no more than five of the 3002 cgMLST loci. These isolates also differed by no more than five nonrepetitive core SNPs. Similar discoveries of transmissions of E. coli between humans and wild birds are described below. We anticipate that large numbers of such previously silent transmission chains will be revealed as EnteroBase is used more extensively.
Case Study 2 illustrates how EnteroBase can facilitate combining reconstructed genomes from metagenomic sequences with draft genomes from cultured strains. In this case, the metagenomes were from ancient tooth pulp that had been enriched for Y. pestis, and the bacterial isolates were modern Y. pestis from a variety of global sources since 1898. The resulting phylogenetic tree (Fig. 5) presents a unique overview of the core genomic diversity over 5000 years of evolution and pandemic spread of plague, which can now be evaluated and used by a broad audience. This tree will be updated at regular intervals as additional genomes or metagenomes become available.
The manual population designations in Figure 5 are largely reflected by HC10 clusters. However, it is uncertain whether the current HierCC clusters would be stable with time because they were based on only 1300 Y. pestis genomes. EnteroBase will therefore maintain these manual annotations in parallel with automated HierCC assignments until a future date when a qualified choice is possible.
Case Study 3 defines the EcoRPlus Collection of 9479 genomes, which represents the genetic diversity of 52,876 genomes. It is a worthy successor of EcoR (Ochman and Selander 1984), which contained 72 representatives of 2600 E. coli strains that had been tested by multilocus enzyme electrophoresis in the early 1980s. The genomic assemblies and known metadata of EcoRPlus are publicly available (http://enterobase.warwick.ac.uk/a/15931) and can serve as a reference set of genomes for future analyses with other methods.
Visual examination of an NJ tree of cgMLST allelic diversity color coded by HierCC HC1100 immediately revealed several discrete E. coli populations that have each been the topics of multiple publications (Fig. 6). These included a primary cause of hemolytic uremic syndrome (O157:H7), a common cause of invasive disease in the elderly (the ST131 Complex), as well as multiple distinct clusters of Shigella that cause dysentery. However, it also contains multiple other discrete clusters of E. coli that are apparently also common causes of global disease in humans and animals, but which have not yet received comparable attention. The annotation of this tree would therefore be a laudable task for the entire scientific community interested in Escherichia. We also note that HierCC is apparently a one stop, complete replacement for haplogroups, Clermont Typing, and ST Complexes, some of whose deficiencies are also illustrated here.
This case study also opened up new perspectives during the review phase of this paper, such as how EnteroBase could be used for the analysis of interhost transmission of antimicrobial resistance (AMR). Seagulls often carry E. coli that are resistant to multiple antibiotics and can transmit those bacteria to other seagulls (Stedt et al. 2014; Ahlstrom et al. 2018, 2019b; Sandegren et al. 2018), including at multiple sites in a small area of Alaska between which seagulls flew on a daily basis (Ahlstrom et al. 2019a). We were therefore not surprised to find that some E. coli isolates from seagulls at those locations were associated within HC5 hierarchical clusters. We then searched for transmissions of HC5 clusters between seagulls and other hosts. EnteroBase contained 406 E. coli genomes from seagulls, distributed over 322 HC5 clusters. Of those clusters, four contained E. coli strains isolated from other hosts (Supplemental Table S5), including chickens, crows, swine, and humans. The dates of isolation of those isolates ranged over about 4 yr, and their geographical locations were separated by long distances: Alaska–New York; Alaska–Michigan; Tasmania–continental Australia. As indicated above, HC5 clusters in Salmonella are associated with recent transmission chains between badgers and across European borders. These additional observations suggest that E. coli from diverse ST Complexes which encode AMR have also been recently transmitted between humans and wild birds and domesticated animals.
This user's guide provides an overview of what EnteroBase can do now. With time, we hope to include additional, currently missing features, such as community annotation of the properties of bacterial populations, predicting antimicrobial resistance/sensitivity, and distributing core pipelines to multiple mirror sites. However, EnteroBase is already able to help a broad community of users with a multitude of tasks for the selected genera it supports. More detailed instructions are available in the online documentation (https://enterobase.readthedocs.io/en/latest/), and questions can be addressed to the support team ([email protected]).
Methods
Isolation of serovar Agama from badgers
Supplemental Figure S5B provides a geographical overview of the area in Woodchester, Gloucestershire, in which badger setts and social groups were investigated in 2006–2007. This area has been subject to a multidecade investigation of badger mobility and patterns of infection with Mycobacterium bovis (McDonald et al. 2018). According to the standard protocol for that study, badgers were subjected to routine capture using steel mesh box traps baited with peanuts, examination under anesthesia, and subsequent release. Fecal samples were cultivated at University of Liverpool after selective enrichment (Rappaport–Vassiliadis broth and semisolid agar), followed by cultivation on MacConkey agar. Lactose-negative colonies that swarmed on Rappaport–Vassiliadis agar but not on nutrient agar, and were catalase-positive and oxidase-negative, were serotyped by slide agglutination tests according to the Kauffmann and White scheme (Issenhuth-Jeanjean et al. 2014). Additional isolates from badgers from the geographical areas in England that are indicated in Supplemental Figure S5D–F were collected during routine investigations of animal disease at the APHA.
Laboratory manipulations and genomic sequencing
At University of Warwick, Salmonella were cultivated, and DNA was purified by automated procedures as described (O'Farrell et al. 2012). Paired-end 150-bp genomic sequencing was performed in multiplexes of 96–192 samples on an Illumina NextSeq 500 using the High Output Kit v2.5 (FC-404-2002) according to the manufacturer's instructions. Other institutions used their own standard procedures. Metadata and features of all 344 genomes in Figure 4 are publicly available in EnteroBase in the workspace “Zhou et al. All Agama strains” (http://enterobase.warwick.ac.uk/a/21320).
Integration of ancient Yersinia pestis genomes in EnteroBase
Metagenomic reads from ancient samples may contain a mixture of sequence reads from the species of interest as well as from genetically similar taxa that represent environmental contamination. To deal with this issue and remove such nonspecific reads after extraction with the EToKi prepare module, the EToKi assemble module can be used to align the extracted reads after comparisons with an ingroup of genomes related to the species of interest and with an outgroup of genomes from other species. In the case of Figure 5, the ingroup consisted of Y. pestis genomes CO92 (2001), Pestoides F, KIM10+ and 91001, and the outgroup consisted of Y. pseudotuberculosis genomes IP32953 and IP31758, Y. similis 228, and Y. enterocolitica 8081. Reads were excluded which had higher alignment scores to the outgroup genomes than to the ingroup genomes. Prior to mapping reads to the Y. pestis reference genome (CO92) (2001), a pseudogenome was created in which all nucleotides were masked to ensure that only nucleotides supported by metagenomic reads would be used for phylogenetic analysis. For the 13 ancient genomes whose publications included complete SNP lists, we unmasked the sites in the pseudogenomes that were included in the published SNP lists. For the other 43 genomes, EToKi was used as in Supplemental Figure S6 to map the filtered metagenomic reads onto the pseudogenome with minimap2 (Li 2018), evaluate them with Pilon (Walker et al. 2014), and unmask sites in the pseudogenome that were covered by three or more reads and had a consensus base that was supported by ≥80% of the mapped reads. All 56 pseudogenomes were uploaded to EnteroBase together with their associated metadata.
Data access
The Illumina sequence reads for 161 new genomes of S. enterica serovar Agama generated in this study have been submitted to the European Nucleotide Archive database (ENA; https://www.ebi.ac.uk/ena) under study accession numbers ERP114376, ERP114456, ERP114871, and ERP115055. The genomic properties, metadata, and accession codes for the 329 genomic assemblies in HC2000_299 are summarized in Supplemental Table S3 and in Online Table 1 (https://wrap.warwick.ac.uk/128112). The metadata, genomic assemblies, and annotations are also available from the publicly available workspace “Zhou et al. All Agama Strains” (http://enterobase.warwick.ac.uk/a/21320). The EToKi package and its documentation are accessible at https://github.com/zheminzhou/EToKi and as Supplemental Code. EnteroBase documentation is accessible at https://enterobase.readthedocs.io/en/latest/. An interactive version of Figure 3 is available at http://enterobase.warwick.ac.uk/a/24006. Trees presented in Figure 4A,B are available separately at http://enterobase.warwick.ac.uk/a/21773/d and http://enterobase.warwick.ac.uk/a/23882/d, respectively. An interactive version of Figure 5 is available at http://enterobase.warwick.ac.uk/a/21977/g. The MicroReact projects of Supplemental Figure S5A,B are available at https://microreact.org/project/t7qlSSslh/3e634888; Supplemental Figure S5C,D at https://microreact.org/project/9XUC7i-Fm/fed65ff5, and Supplemental Figure S5E,F at https://microreact.org/project/XaJm1cNjY/69748fe3. The tree shown in Figure 6 as well as Supplemental Figures S7, S8 are available at http://enterobase.warwick.ac.uk/a/15981.
Agama Study Group
Derek Brown,3 Marie Chattaway,4 Tim Dallman,4 Richard Delahay,5 Christian Kornschober,6 Ariane Pietzka,6 Burkhard Malorny,7 Liljana Petrovska,8 Rob Davies,8 Andy Robertson,9 William Tyne,10 François-Xavier Weill,11 Marie Accou-Demartin,11 Nicola Williams,12
Acknowledgments
EnteroBase development was funded by the Biotechnology and Biological Sciences Research Council (BB/L020319/1) and the Wellcome Trust (202792/Z/16/Z). We thank Niall Delappe and Martin Cormican, Salmonella Reference Laboratory, Galway, Ireland, for sharing strains and data, and Nina Luhmann and Jane Charlesworth for critical comments on the text.
Author contributions: M.A. wrote the manuscript with the help of all other authors. Z.Z., N.-F.A., K.M., and Y.F. were responsible for the development of EnteroBase under the guidance of M.A. N.-F.A. and K.M. were responsible for the online manual. Analyses were performed and figures were drawn by Z.Z. and N.-F.A. under the guidance of M.A. The Agama Study Group provided information, bacterial strains, DNAs, and genomic sequences from Agama isolates from all over Europe and was involved in writing the manuscript and evaluating the conclusions.
Notes
[1] Supplementary material [Supplemental material is available for this article.]
[2] Article published online before print. Article, supplemental material, and publication date are at http://www.genome.org/cgi/doi/10.1101/gr.251678.119.
[3] Freely available online through the Genome Research Open Access option.
[4] Scottish Salmonella Reference Laboratory, Glasgow G31 2ER, UK
[5] Public Health England (PHE), Colindale, London NW9 5EQ, UK
[6] National Wildlife Management Centre, APHA, Sand Hutton, York YO41 1LZ, UK
[7] Austrian Agency for Health and Food Safety (AGES), Institute for Medical Microbiology and Hygiene, 8010 Graz, Austria
[8] German Federal Institute for Risk Assessement, D-10589 Berlin, Germany (Study Centre for Genome Sequencing and Analysis)
[9] Animal and Plant Health Agency (APHA), Addlestone KT15 3NB, UK
[10] Environment and Sustainability Institute, University of Exeter, Penryn TR10 9FE, UK
[11] Warwick Medical School, University of Warwick, Coventry CV4 7AL, UK
[12] Institut Pasteur, 75724 Paris cedex, France
[13] Department of Epidemiology and Population Health, Institute of Infection and Global Health, University of Liverpool, Neston CH64 7TE, UK
References
- ↵Achtman M. 2016. How old are bacterial pathogens? Proc Biol Sci 283: 20160990. 10.1098/rspb.2016.0990
- ↵Achtman M, Zhou Z. 2014. Distinct genealogies for plasmids and chromosome. PLoS Genet 10: e1004874. 10.1371/journal.pgen.1004874
- ↵Achtman M, Zhou Z. 2019. Analysis of the human oral microbiome from modern and historical samples with SPARSE and EToKi. bioRxiv 10.1101/842542
- ↵Achtman M, Wain J, Weill FX, Nair S, Zhou Z, Sangal V, Krauland MG, Hale JL, Harbottle H, Uesbeck A, 2012. Multilocus sequence typing as a replacement for serotyping in Salmonella enterica. PLoS Pathog 8: e1002776. 10.1371/journal.ppat.1002776
- ↵Ahlstrom CA, Bonnedahl J, Woksepp H, Hernandez J, Olsen B, Ramey AM. 2018. Acquisition and dissemination of cephalosporin-resistant E. coli in migratory birds sampled at an Alaska landfill as inferred through genomic analysis. Sci Rep 8: 7361. 10.1038/s41598-018-25474-w
- ↵Ahlstrom CA, Bonnedahl J, Woksepp H, Hernandez J, Reed JA, Tibbitts L, Olsen B, Douglas DC, Ramey AM. 2019a. Satellite tracking of gulls and genomic characterization of faecal bacteria reveals environmentally mediated acquisition and dispersal of antimicrobial-resistant Escherichia coli on the Kenai Peninsula, Alaska. Mol Ecol 28: 2531–2545. 10.1111/mec.15101
- ↵Ahlstrom CA, Ramey AM, Woksepp H, Bonnedahl J. 2019b. Repeated detection of carbapenemase-producing Escherichia coli in gulls inhabiting Alaska. Antimicrob Agents Chemother 63: e00758-19. 10.1128/AAC.00758-19
- ↵Alikhan NF, Zhou Z, Sergeant MJ, Achtman M. 2018. A genomic overview of the population structure of Salmonella. PLoS Genet 14: e1007261. 10.1371/journal.pgen.1007261
- ↵Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ. 1990. Basic local alignment search tool. J Mol Biol 215: 403–410. 10.1016/S0022-2836(05)80360-2
- ↵Argimón S, Abudahab K, Goater RJ, Fedosejev A, Bhai J, Glasner C, Feil EJ, Holden MT, Yeats CA, Grundmann H, 2016. Microreact: visualizing and sharing data for genomic epidemiology and phylogeography. Microb Genom 2: e000093. 10.1099/mgen.0.000093
- ↵Ashton PM, Owen SV, Kaindama L, Rowe WPM, Lane CR, Larkin L, Nair S, Jenkins C, de Pinna EM, Feasey NA, 2017. Public health surveillance in the UK revolutionises our understanding of the invasive Salmonella Typhimurium epidemic in Africa. Genome Med 9: 92. 10.1186/s13073-017-0480-7
- ↵Beghain J, Bridier-Nahmias A, Le NH, Denamur E, Clermont O. 2018. ClermonTyping: an easy-to-use and accurate in silico method for Escherichia genus strain phylotyping. Microb Genom 4: e000192. 10.1099/mgen.0.000192
- ↵Bird S, Klein E, Loper E. 2009. Natural language processing with Python: analyzing text with the Natural Language Toolkit, 1st ed. O'Reilly Media, Sebastopol, CA.
- ↵Bos KI, Schuenemann VJ, Golding GB, Burbano HA, Waglechner N, Coombes BK, McPhee JB, Dewitte SN, Meyer M, Schmedes S, 2011. A draft genome of Yersinia pestis from victims of the Black Death. Nature 478: 506–510. 10.1038/nature10549
- ↵Bos KI, Herbig A, Sahl J, Waglechner N, Fourment M, Forrest SA, Klunk J, Schuenemann VJ, Poinar D, Kuch M, 2016. Eighteenth century Yersinia pestis genomes reveal the long-term persistence of an historical plague focus. eLife 5: e12994. 10.7554/eLife.12994
- ↵Clark K, Karsch-Mizrachi I, Lipman DJ, Ostell J, Sayers EW. 2016. GenBank. Nucleic Acids Res 44: D67–D72. 10.1093/nar/gkv1276
- ↵Clermont O, Christenson JK, Denamur E, Gordon DM. 2013. The Clermont Escherichia coli phylo-typing method revisited: improvement of specificity and detection of new phylo-groups. Environ Microbiol Rep 5: 58–65. 10.1111/1758-2229.12019
- ↵Connor TR, Owen SV, Langridge G, Connell S, Nair S, Reuter S, Dallman TJ, Corander J, Tabing KC, Le Hello S, 2016. What's in a name? Species-wide whole-genome sequencing resolves invasive and noninvasive lineages of Salmonella enterica serotype Paratyphi B. MBio 7: e00527-16. 10.1128/mBio.00527-16
- ↵Cui Y, Yu C, Yan Y, Li D, Li Y, Jombart T, Weinert LA, Wang Z, Guo Z, Xu L, 2013. Historical variations in mutation rate in an epidemic pathogen, Yersinia pestis. Proc Natl Acad Sci 110: 577–582. 10.1073/pnas.1205750110
- ↵Dallman T, Inns T, Jombart T, Ashton P, Loman N, Chatt C, Messelhaeusser U, Rabsch W, Simon S, Nikisins S, 2016. Phylogenetic structure of European Salmonella Enteritidis outbreak correlates with national and international egg distribution network. Microb Genom 2: e000070. 10.1099/mgen.0.000070
- ↵Damgaard PB, Marchi N, Rasmussen S, Peyrot M, Renaud G, Korneliussen T, Moreno-Mayar JV, Pedersen MW, Goldberg A, Usmanova E, 2018. 137 ancient human genomes from across the Eurasian steppes. Nature 557: 369–374. 10.1038/s41586-018-0094-2
- ↵Diemert S, Yan T. 2019. Clinically unreported salmonellosis outbreak detected via comparative genomic analysis of municipal wastewater Salmonella isolates. Appl Environ Microbiol 85: e00139-19. 10.1128/AEM.00139-19
- ↵Edgar RC. 2010. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26: 2460–2461. 10.1093/bioinformatics/btq461
- ↵Eppinger M, Mammel MK, LeClerc JE, Ravel J, Cebula TA. 2011a. Genome signatures of Escherichia coli O157:H7 from the bovine host reservoir. Appl Environ Microbiol 77: 2916–2925. 10.1128/AEM.02554-10
- ↵Eppinger M, Mammel MK, LeClerc JE, Ravel J, Cebula TA. 2011b. Genomic anatomy of Escherichia coli O157:H7 outbreaks. Proc Natl Acad Sci 108: 20142–20147. 10.1073/pnas.1107176108
- ↵Eroshenko GA, Nosov NY, Krasnov YM, Oglodin YG, Kukleva LM, Guseva NP, Kuznetsov AA, Abdikarimov ST, Dzhaparova AK, Kutyrev VV. 2017. Yersinia pestis strains of ancient phylogenetic branch 0.ANT are widely spread in the high-mountain plague foci of Kyrgyzstan. PLoS One 12: e0187230. 10.1371/journal.pone.0187230
- ↵Feil EJ, Li BC, Aanensen DM, Hanage WP, Spratt BG. 2004. eBURST: inferring patterns of evolutionary descent among clusters of related bacterial genotypes from multilocus sequence typing data. J Bacteriol 186: 1518–1530. 10.1128/JB.186.5.1518-1530.2004
- ↵Feldman M, Harbeck M, Keller M, Spyrou MA, Rott A, Trautmann B, Scholz HC, Päffgen B, Peters J, McCormick M, 2016. A high-coverage Yersinia pestis genome from a sixth-century Justinianic plague victim. Mol Biol Evol 33: 2911–2923. 10.1093/molbev/msw170
- ↵Frentrup M, Zhou Z, Steglich M, Meier-Kolthoff JP, Göker M, Riedel T, Bunk B, Spröer C, Overmann J, Blaschitz M, 2019. Global genomic population structure of Clostridioides difficile. bioRxiv 10.1101/727230
- ↵Gordon DM, Geyik S, Clermont O, O'Brien CL, Huang S, Abayasekara C, Rajesh A, Kennedy K, Collignon P, Pavli P, 2017. Fine-scale structure analysis shows epidemic patterns of clonal complex 95, a cosmopolitan Escherichia coli lineage responsible for extraintestinal infection. mSphere 2: e00168-17. 10.1128/mSphere.00168-17
- ↵Green MH. 2018. Putting Africa on the Black Death map: narratives from genetics and history. Afriques [Online] 9. 10.4000/afriques.2125
- Griffiths D, Fawley W, Kachrimanidou M, Bowden R, Crook DW, Fung R, Golubchik T, Harding RM, Jeffery KJ, Jolley KA, 2010. Multilocus sequence typing of Clostridium difficile. J Clin Microbiol 48: 770–778. 10.1128/JCM.01796-09
- ↵Guibourdenche M, Roggentin P, Mikoleit M, Fields PI, Bockemühl J, Grimont PA, Weill FX. 2010. Supplement 2003–2007 (No. 47) to the White-Kauffmann-Le Minor scheme. Res Microbiol 161: 26–29. 10.1016/j.resmic.2009.10.002
- ↵Haley BJ, Kim SW, Haendiges J, Keller E, Torpey D, Kim A, Crocker K, Myers RA, Van Kessel JAS. 2019. Salmonella enterica serovar Kentucky recovered from human clinical cases in Maryland, USA (2011–2015). Zoonoses Public Health 66: 382–392. 10.1111/zph.12571
- Hall M, Chattaway MA, Reuter S, Savin C, Strauch E, Carniel E, Connor T, Van Damme I, Rajakaruna L, Rajendram D, 2015. Use of whole-genus genome sequence data to develop a multilocus sequence typing tool that accurately identifies Yersinia isolates to the species and subspecies levels. J Clin Microbiol 53: 35–42. 10.1128/JCM.02395-14
- ↵Issenhuth-Jeanjean S, Roggentin P, Mikoleit M, Guibourdenche M, De Pinna E, Nair S, Fields PI, Weill FX. 2014. Supplement 2008–2010 (No. 48) to the White-Kauffmann-Le Minor scheme. Res Microbiol 165: 526–530. 10.1016/j.resmic.2014.07.004
- ↵Johnson TJ, Elnekave E, Miller EA, Munoz-Aguayo J, Flores FC, Johnston B, Nielson DW, Logue CM, Johnson JR. 2019. Phylogenomic analysis of extraintestinal pathogenic Escherichia coli sequence type 1193, an emerging multidrug-resistant clonal group. Antimicrob Agents Chemother 63: e01913-18. 10.1128/AAC.01913-18
- ↵Jolley KA, Bliss CM, Bennett JS, Bratcher HB, Brehony C, Colles FM, Wimalarathna H, Harrison OB, Sheppard SK, Cody AJ, 2012. Ribosomal multilocus sequence typing: universal characterization of bacteria from domain to strain. Microbiology 158: 1005–1015. 10.1099/mic.0.055459-0
- ↵Jones G, Lefèvre S, Donguy MP, Nisavanh A, Terpant G, Fougère E, Vaissière E, Guinard A, Mailles A, De Valk H, 2019a. Outbreak of Shiga toxin-producing Escherichia coli (STEC) O26 paediatric haemolytic uraemic syndrome (HUS) cases associated with the consumption of soft raw cow's milk cheeses, France, March to May 2019. Euro Surveill 24: 1900305. 10.2807/1560-7917.ES.2019.24.22.1900305
- ↵Jones G, Pardos de la Gandaro M, Herrera-Leon L, Herrera-Leon S, Varela Martinez C, Hureaux-Roy R, Abdallah Y, Nisavanh A, Fabre L, Renaudat C, 2019b. Outbreak of Salmonella enterica serotype Poona in infants linked to persistent Salmonella contamination in an infant formula manufacturing facility, France, August 2018 to February 2019. Euro Surveill 24: 1900161. 10.2807/1560-7917.ES.2019.24.13.1900161
- ↵Keller M, Spyrou MA, Scheib CL, Neumann GU, Kröpelin A, Haas-Gebhard B, Päffgen B, Haberstroh J, Ribera IL, Raynaud C, 2019. Ancient Yersinia pestis genomes from across Western Europe reveal early diversification during the First Pandemic (541–750). Proc Natl Acad Sci 116: 12363–12372. 10.1073/pnas.1820447116
- Kidgell C, Reichard U, Wain J, Linz B, Torpdahl M, Dougan G, Achtman M. 2002. Salmonella typhi, the causative agent of typhoid fever, is approximately 50,000 years old. Infect Genet Evol 2: 39–45. 10.1016/S1567-1348(02)00089-8
- ↵Kutyrev VV, Eroshenko GA, Motin VL, Nosov NY, Krasnov JM, Kukleva LM, Nikiforov KA, Al'khova ZV, Oglodin EG, Guseva NP. 2018. Phylogeny and classification of Yersinia pestis through the lens of strains from the plague foci of Commonwealth of Independent States. Front Microbiol 9: 1106. 10.3389/fmicb.2018.01106
- ↵Langridge GC, Fookes M, Connor TR, Feltwell T, Feasey N, Parsons BN, Seth-Smith HM, Barquist L, Stedman A, Humphrey T, 2015. Patterns of genome evolution that have accompanied host adaptation in Salmonella. Proc Natl Acad Sci 112: 863–868. 10.1073/pnas.1416707112
- ↵Laukkanen-Ninios R, Didelot X, Jolley KA, Morelli G, Sangal V, Kristo P, Brehony C, Imori PF, Fukushima H, Siitonen A, 2011. Population structure of the Yersinia pseudotuberculosis complex according to multilocus sequence typing. Environ Microbiol 13: 3114–3127. 10.1111/j.1462-2920.2011.02588.x
- ↵Li H. 2018. Minimap2: pairwise alignment for nucleotide sequences. Bioinformatics 34: 3094–3100. 10.1093/bioinformatics/bty191
- ↵Liu S, Jin D, Lan R, Wang Y, Meng Q, Dai H, Lu S, Hu S, Xu J. 2015. Escherichia marmotae sp. nov., isolated from faeces of Marmota himalayana. Int J Syst Evol Microbiol 65: 2130–2134. 10.1099/ijs.0.000228
- ↵Liu CM, Stegger M, Aziz M, Johnson TJ, Waits K, Nordstrom L, Gauld L, Weaver B, Rolland D, Statham S, 2018. Escherichia coli ST131-H22 as a foodborne uropathogen. MBio 9: e00470-18. 10.1128/mBio.00470-18
- ↵Luo C, Walk ST, Gordon DM, Feldgarden M, Tiedje JM, Konstantinidis KT. 2011. Genome sequencing of environmental Escherichia coli expands understanding of the ecology and speciation of the model bacterial species. Proc Natl Acad Sci 108: 7200–7205. 10.1073/pnas.1015622108
- ↵Maiden MCJ, Bygraves JA, Feil E, Morelli G, Russell JE, Urwin R, Zhang Q, Zhou J, Zurth K, Caugant DA, 1998. Multilocus sequence typing: a portable approach to the identification of clones within populations of pathogenic microorganisms. Proc Natl Acad Sci 95: 3140–3145. 10.1073/pnas.95.6.3140
- Maiden MC, van Rensburg MJ, Bray JE, Earle SG, Ford SA, Jolley KA, McCarthy ND. 2013. MLST revisited: the gene-by-gene approach to bacterial genomics. Nat Rev Microbiol 11: 728–736. 10.1038/nrmicro3093
- ↵Margaryan A, Hansen HB, Rasmussen S, Sikora M, Moiseyev V, Khoklov A, Epimakhov A, Yepiskoposyan L, Kriiska A, Varul L, 2018. Ancient pathogen DNA in human teeth and petrous bones. Ecol Evol 8: 3534–3542. 10.1002/ece3.3924
- ↵McDonald JL, Robertson A, Silk MJ. 2018. Wildlife disease ecology from the individual to the population: insights from a long-term study of a naturally infected European badger population. J Anim Ecol 87: 101–112. 10.1111/1365-2656.12743
- Mellmann A, Harmsen D, Cummings CA, Zentz EB, Leopold SR, Rico A, Prior K, Szczepanowski R, Ji Y, Zhang W, 2011. Prospective genomic characterization of the German enterohemorrhagic Escherichia coli O104:H4 outbreak by rapid next generation sequencing technology. PLoS One 6: e22751. 10.1371/journal.pone.0022751
- ↵Miller EA, Elnekave E, Figueroa CF, Johnson A, Kearney A, Aguayo JM, Tagg K, Tschetter L, Weber B, Nadon C, 2019. Emergence of a novel Salmonella enterica serotype Reading clone is linked to its expansion in commercial turkey production, resulting in unanticipated human illness in North America. bioRxiv 10.1101/855734
- ↵Morelli G, Song Y, Mazzoni CJ, Eppinger M, Roumagnac P, Wagner DM, Feldkamp M, Kusecek B, Vogler AJ, Li Y, 2010. Yersinia pestis genome sequencing identifies patterns of global phylogenetic diversity. Nature Genet 42: 1140–1143. 10.1038/ng.705
- ↵Namouchi A, Guellil M, Kersten O, Hänsch S, Ottoni C, Schmid BV, Pacciani E, Quaglia L, Vermunt M, Bauer EL, 2018. Integrative approach using Yersinia pestis genomes to revisit the historical landscape of plague during the Medieval Period. Proc Natl Acad Sci 115: E11790–E11797. 10.1073/pnas.1812865115
- ↵Newell DG, La Ragione RM. 2018. Enterohaemorrhagic and other Shiga toxin-producing Escherichia coli (STEC): Where are we now regarding diagnostics and control strategies? Transbound Emerg Dis 65: 49–71. 10.1111/tbed.12789
- ↵Numberger D, Riedel T, McEwen G, Nübel U, Frentrup M, Schober I, Bunk B, Spröer C, Overmann J, Grossart HP, 2019. Genomic analysis of three Clostridioides difficile isolates from urban water sources. Anaerobe 56: 22–26. 10.1016/j.anaerobe.2019.01.002
- ↵Ochman H, Selander RK. 1984. Standard reference strains of Escherichia coli from natural populations. J Bacteriol 157: 690–693.
- ↵O'Farrell B, Haase JK, Velayudhan V, Murphy RA, Achtman M. 2012. Transforming microbial genotyping: a robotic pipeline for genotyping bacterial strains. PLoS One 7: e48022. 10.1371/journal.pone.0048022
- ↵Parkhill J, Wren BW, Thomson NR, Titball RW, Holden MT, Prentice MB, Sebaihia M, James KD, Churcher C, Mungall KL, 2001. Genome sequence of Yersinia pestis, the causative agent of plague. Nature 413: 523–527. 10.1038/35097083
- ↵Pupo GM, Lan R, Reeves PR. 2000. Multiple independent origins of Shigella clones of Escherichia coli and convergent evolution of many of their characteristics. Proc Natl Acad Sci 97: 10567–10572. 10.1073/pnas.180094797
- ↵Rasmussen S, Allentoft ME, Nielsen K, Orlando L, Sikora M, Sjögren KG, Pedersen AG, Schubert M, Van DA, Kapel CM, 2015. Early divergent strains of Yersinia pestis in Eurasia 5,000 years ago. Cell 163: 571–582. 10.1016/j.cell.2015.10.009
- ↵Robertson J, Yoshida C, Kruczkiewicz P, Nadon C, Nichani A, Taboada EN, Nash JHE. 2018. Comprehensive assessment of the quality of Salmonella whole genome sequence data available in public sequence databases using the Salmonella in silico Typing Resource (SISTR). Microb Genom 4: e000151. 10.1099/mgen.0.000151
- ↵Roer L, Tchesnokova V, Allesøe R, Muradova M, Chattopadhyay S, Ahrenfeldt J, Thomsen MCF, Lund O, Hansen F, Hammerum AM, 2017. Development of a web tool for Escherichia coli subtyping based on fimH alleles. J Clin Microbiol 55: 2538–2543. 10.1128/JCM.00737-17
- ↵Rogers LM, Delahay R, Cheeseman CL, Langton S, Smith GC, Clifton-Hadley RS. 1998. Movement of badgers (Meles meles) in a high-density population: individual, population and disease effects. Proc Biol Sci 265: 1269–1276. 10.1098/rspb.1998.0429
- ↵Sanaa M, Pouillot R, Vega FG, Strain E, Van Doren JM. 2019. GenomeGraphR: a user-friendly open-source web application for foodborne pathogen whole genome sequencing data integration, analysis, and visualization. PLoS One 14: e0213039. 10.1371/journal.pone.0213039
- ↵Sandegren L, Stedt J, Lustig U, Bonnedahl J, Andersson DI, Järhult JD. 2018. Long-term carriage and rapid transmission of extended spectrum β-lactamase-producing E. coli within a flock of Mallards in the absence of antibiotic selection. Environ Microbiol Rep 10: 576–582. 10.1111/1758-2229.12681
- ↵Selander RK, Caugant DA, Whittam TS. 1987. Genetic structure and variation in natural populations of Escherichia coli. In Escherichia coli and Salmonella typhimurium cellular and molecular biology (ed. Neidhardt FC, ), Vol. II, pp. 1625–1648. American Society for Microbiology, Washington, DC.
- ↵Simonsen M, Mailund T, Pedersen CNS. 2011. Inference of large phylogenies using neighbour-joining. In Biomedical Engineering Systems and Technologies: 3rd International Joint Conference, BIOSTEC 2010, pp. 334–344. Springer-Verlag, Berlin, Germany.
- ↵Spyrou MA, Tukhbatova RI, Feldman M, Drath J, Kacki S, Beltran de HJ, Arnold S, Sitdikov AG, Castex D, Wahl J, 2016. Historical Y. pestis genomes reveal the European Black Death as the source of ancient and modern plague pandemics. Cell Host Microbe 19: 874–881. 10.1016/j.chom.2016.05.012
- ↵Spyrou MA, Tukhbatova RI, Wang CC, Valtueña AA, Lankapalli AK, Kondrashin VV, Tsybin VA, Khokhlov A, Kühnert D, Herbig A, 2018. Analysis of 3800-year-old Yersinia pestis genomes suggests Bronze Age origin for bubonic plague. Nat Commun 9: 2234. 10.1038/s41467-018-04550-9
- ↵Spyrou MA, Keller M, Tukhbatova RI, Scheib CL, Nelson EA, Andrades VA, Neumann GU, Walker D, Alterauge A, Carty N, 2019. Phylogeography of the second plague pandemic revealed through analysis of historical Yersinia pestis genomes. Nat Commun 10: 4470. 10.1038/s41467-019-12154-0
- ↵Stamatakis A. 2014. RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 30: 1312–1313. 10.1093/bioinformatics/btu033
- ↵Stedt J, Bonnedahl J, Hernandez J, McMahon BJ, Hasan B, Olsen B, Drobni M, Waldenström J. 2014. Antibiotic resistance patterns in Escherichia coli from gulls in nine European countries. Infect Ecol Epidemiol 4: 21565. 10.3402/iee.v4.21565
- ↵Stoesser N, Sheppard AE, Pankhurst L, De Maio N, Moore CE, Sebra R, Turner P, Anson LW, Kasarskis A, Batty EM, 2016. Evolutionary history of the global emergence of the Escherichia coli epidemic clone ST131. MBio 7: e02162. 10.1128/mBio.02162-15
- ↵van der Putten BCL, Matamoros S, COMBAT Consortium, Schultsz C. 2019. Genomic evidence for revising the Escherichia genus and description of Escherichia ruysiae sp. nov. bioRxiv 10.1101/781724
- ↵Wagner DM, Klunk J, Harbeck M, Devault A, Waglechner N, Sahl JW, Enk J, Birdsell DN, Kuch M, Lumibao C, 2014. Yersinia pestis and the plague of Justinian 541–543 AD: a genomic analysis. Lancet Infect Dis 14: 319–326. 10.1016/S1473-3099(13)70323-2
- ↵Waldram A, Dolan G, Ashton PM, Jenkins C, Dallman TJ. 2018. Epidemiological analysis of Salmonella clusters identified by whole genome sequencing, England and Wales 2014. Food Microbiol 71: 39–45. 10.1016/j.fm.2017.02.012
- ↵Walk ST, Alm EW, Gordon DM, Ram JL, Toranzos GA, Tiedje JM, Whittam TS. 2009. Cryptic lineages of the genus Escherichia. Appl Environ Microbiol 75: 6534–6544. 10.1128/AEM.01262-09
- ↵Walker BJ, Abeel T, Shea T, Priest M, Abouelleil A, Sakthikumar S, Cuomo CA, Zeng Q, Wortman J, Young SK, 2014. Pilon: an integrated tool for comprehensive microbial variant detection and genome assembly improvement. PLoS One 9: e112963. 10.1371/journal.pone.0112963
- ↵Waters NR, Abram F, Brennan F, Holmes A, Pritchard L. 2018. Easily phylotyping E. coli via the EzClermont web app and command-line tool. bioRxiv 10.1101/317610
- ↵Wilson JS, Hazel SM, Williams NJ, Phiri A, French NP, Hart CA. 2003. Nontyphoidal salmonellae in United Kingdom badgers: prevalence and spatial distribution. Appl Environ Microbiol 69: 4312–4315. 10.1128/AEM.69.7.4312-4315.2003
- ↵Wirth T, Falush D, Lan R, Colles F, Mensa P, Wieler LH, Karch H, Reeves PR, Maiden MC, Ochman H, 2006. Sex and virulence in Escherichia coli: an evolutionary perspective. Mol Microbiol 60: 1136–1151. 10.1111/j.1365-2958.2006.05172.x
- ↵Wirth T, Morelli G, Kusecek B, Van Belkum A, van der Schee C, Meyer A, Achtman M. 2007. The rise and spread of a new pathogen: seroresistant Moraxella catarrhalis. Genome Res 17: 1647–1656. 10.1101/gr.6122607
- ↵Wong VK, Baker S, Connor TR, Pickard D, Page AJ, Dave J, Murphy N, Holliman R, Sefton A, Millar M, 2016. An extended genotyping framework for Salmonella enterica serovar Typhi, the cause of human typhoid. Nat Commun 7: 12827. 10.1038/ncomms12827
- ↵Worley J, Meng J, Allard MW, Brown EW, Timme RE. 2018. Salmonella enterica phylogeny based on whole-genome sequencing reveals two new clades and novel patterns of horizontally acquired genetic elements. MBio 9: e02303-18. 10.1128/mBio.02303-18
- ↵Wray C, Baker K, Gallagher J, Naylor P. 1977. Salmonella infection in badgers in the South West of England. Br Vet J 133: 526–529. 10.1016/S0007-1935(17)33996-9
- ↵Yoshida CE, Kruczkiewicz P, Laing CR, Lingohr EJ, Gannon VP, Nash JH, Taboada EN. 2016. The Salmonella In Silico Typing Resource (SISTR): an open web-accessible tool for rapidly typing and subtyping draft Salmonella genome assemblies. PLoS One 11: e0147101. 10.1371/journal.pone.0147101
- ↵Zhang S, Den-Bakker HC, Li S, Chen J, Dinsmore BA, Lane C, Lauer AC, Fields PI, Deng X. 2019. SeqSero2: rapid and improved Salmonella serotype determination using whole genome sequencing data. Appl Environ Microbiol 85: e01746-19. 10.1128/AEM.01746-19
- ↵Zhou Z, McCann A, Litrup E, Murphy R, Cormican M, Fanning S, Brown D, Guttman DS, Brisse S, Achtman M. 2013. Neutral genomic microevolution of a recently emerged pathogen, Salmonella enterica serovar Agona. PLoS Genet 9: e1003471. 10.1371/journal.pgen.1003471
- ↵Zhou Z, McCann A, Weill FX, Blin C, Nair S, Wain J, Dougan G, Achtman M. 2014. Transient Darwinian selection in Salmonella enterica serovar Paratyphi A during 450 years of global spread of enteric fever. Proc Natl Acad Sci 111: 12199–12204. 10.1073/pnas.1411012111
- ↵Zhou Z, Alikhan N-F, Sergeant MJ, Luhmann N, Vaz C, Francisco AP, Carriço JA, Achtman M. 2018a. GrapeTree: visualization of core genomic relationships among 100,000 bacterial pathogens. Genome Res 28: 1395–1404. 10.1101/gr.232397.117
- ↵Zhou Z, Luhmann N, Alikhan NF, Quince C, Achtman M. 2018b. Accurate reconstruction of microbial strains from metagenomic sequencing using representative reference genomes. In RECOMB 2018, 225–240. Springer, Cham, Switzerland.
- ↵Zhou Z, Lundstrøm I, Tran-Dien A, Duchêne S, Alikhan NF, Sergeant MJ, Langridge G, Fokatis AK, Nair S, Stenøien HK, 2018c. Pan-genome analysis of ancient and modern Salmonella enterica demonstrates genomic stability of the invasive Para C lineage for millennia. Curr Biol 28: 2420–2428.e10. 10.1016/j.cub.2018.05.058